Our story with calcium hydrogen phosphate stretches back to the early days of industrial chemistry. Before the digital age, producers and researchers set out to refine basic calcium phosphates from naturally occurring minerals. The fertilizer revolution in agriculture brought our focus to calcium hydrogen phosphate, particularly as superphosphate production kicked off in the nineteenth century. We have always found value in the way collaboration among farmers, scientists, and chemical engineers set the stage for reliable, high-purity phosphate salts. Production improvements emerged over decades as glassware gave way to continuous reactors, and batch inconsistencies shifted toward dependable bulk output. The changes in process design respond directly to increased demand in agriculture, animal nutrition, and pharmaceutical specialties, so our own factory lines bear the marks of years of tweaks, modifications, and regulatory advances.
Our batches of calcium hydrogen phosphate, known in the industry as dicalcium phosphate or DCP, support a variety of sectors. Most folks recognize it in the context of feed or food additives, but it also fills prescription tablets, dental polishing powders, and water treatment systems. This adaptability increases the pressure on us to maintain batch consistency, particle size range, and effective solubility, especially for tablet manufacturers who need every granule to behave predictably during compaction and dissolution. Commercially, our product goes by several chemical aliases: calcium hydrogen phosphate, dicalcium orthophosphate, or E341(ii) for those in the food space.
Technical personnel working in our plants handle a fine, odorless powder, white and chalky, flowing easily on the line when conditions are right. Moisture management remains a critical control point; clumping or caking signals off-spec performance, whether headed for livestock feed or tablet press. Solubility in water is modest and pH-neutral environments keep it from turning into other phosphate forms. Chemically, each molecule contains pure calcium and orthophosphate anions, with a crystal structure (most commonly in the dihydrate form) that distinguishes itself from its mono- or tricalcium cousins. Crystallinity, density, and bulk behavior matter for downstream handling, and small tweaks to humidity or process duration shift these properties, so our QC lab tracks each lot stringently.
We base our technical specifications on relevant food, feed, or pharma standards to meet traceability requirements and quality benchmarks. Each label reflects the actual calcium and phosphorus contents, which often hover around 23% calcium and 18% phosphorus by weight, with moisture content tightly managed to prevent over-drying or excess free water. Impurities such as heavy metals and fluorine see close monitoring, especially for animal or human consumption. Up-to-date labeling, with QR-codes or batch numbers, helps downstream users access origin, production date, and related compliance data.
Most of our commercial output comes from combining naturally sourced calcium carbonate (usually from limestone) with phosphoric acid under precisely managed pH and temperature. We employ reactors lined with corrosion-resistant materials—phosphoric acid is aggressive, and material compatibility is non-negotiable. Careful control of agitation, reaction time, and temperature ensures the right hydration state and particle character. Filtration and drying follow, with residual acidified water recycled back into the process. Fine-tuning yields a product tailored to each customer’s demand, whether for free-flowing powders or compactable granules. Byproducts, primarily calcium sulfate, also find use in other industries, enabling us to minimize waste.
Our technical teams often adjust process steps to prepare alternative grades or specialty modifications. By varying reaction conditions, we can lean the process toward monohydrate or trihydrate forms or produce anhydrous grades through controlled drying steps. These variants affect solubility and compaction properties—critical in tablet manufacturing or as flow aids. Phosphate rocks offer another production route, though high-purity demands steer us toward acidulation in most food and pharma contexts. Our R&D team explores co-precipitation with trace minerals, enhancing animal feed efficiency or supplement bioavailability for human nutrition.
Customers typically ask for “DCP” or “Dicalcium Phosphate,” sometimes referencing the CAS number for clarity. Regulatory documents reference “Calcium Hydrogen Phosphate,” and in the context of food, it appears as E341(ii) in European labeling. Variants include dicalcium orthophosphate, calcium phosphate dibasic, and secondary calcium phosphate. Knowing the difference between mono-, di-, and tricalcium phosphate matters greatly, especially in applications requiring a particular calcium-to-phosphorus ratio. Our documentation always clarifies specification and hydration state, short-circuiting confusion in bulk orders or export paperwork.
Handling large quantities of calcium hydrogen phosphate demands rigorous controls. Dust inhalation remains the main worker exposure risk, so we maintain advanced extraction and filtration systems at production points. Operators wear dust masks and gloves, not because the material is particularly toxic, but to avoid cumulative respiratory irritation and to maintain clean working conditions. Storage in moisture-controlled environments prevents caking and degradation. Routine safety drills prepare staff for accidental spills, even though the product itself poses low acute hazard. Compliance with REACH, FDA, or domestic food law ensures downstream users receive material without concerning contaminants or off-spec compositions. Each safety data sheet (SDS) reflects real incident history, not just theoretical hazards.
The bulk of our calcium hydrogen phosphate ships to compound feed manufacturers, who rely on it to balance mineral nutrition for swine, cattle, and poultry. Tablet and capsule formulas in the pharmaceutical industry demand precise particle sizing and solubility curves to ensure reliable dissolution in the digestive tract. Food processors use it as a leavening agent and anti-caking aid, with requirements for taste neutrality and whiteness. Toothpaste and dental powder use appreciate its mild abrasiveness without enamel damage, which derives from those specific crystalline properties achieved on our lines. For water treatment, calcium hydrogen phosphate helps buffer pH and precipitate metals, though these applications demand a different purity tier. Each sector drives us to invest in process consistency and keep an open dialogue with customers facing evolving requirements.
Our R&D efforts follow trends in both process optimization and customer application. Work continues on improving precipitation techniques to enhance yield and purity, and to reduce energy consumption during drying and granulation. We closely monitor advances in mineral bioavailability, with particular attention to micronutrient fortification for both human food supplements and animal nutrition. Collaborative projects with universities and feed formulators take up much of our pilot capacity, as new evidence emerges around trace mineral interactions or improved phosphorus utilization. Research into greener production—using alternative acids, renewable energy, and waste valorization—underscores our strategy meetings. Fast turnaround times for pilot batches let us address high-value specialty requests, especially in pharma and biotech.
Calcium hydrogen phosphate sits low on the hazard scale for acute and chronic toxicity. We have supported third-party studies that confirm its safety for use in food and feed, provided manufacturing avoids introduction of toxic metal contaminants. Chronic overconsumption can lead to mineral imbalance, so regulatory tolerances serve as our production benchmarks. Our internal and external tests show no carcinogenic or mutagenic effect, but we continuously screen for emerging risks tied to nanoparticle inhalation or unusual byproduct contamination. Environmental research tracks long-term aquatic impacts of effluents from bulk users; as a manufacturer, we contribute data to help water regulators verify minimal risk to aquatic life from approved discharges.
Looking ahead, our sector faces both pressure and opportunity. Decarbonization initiatives push us to adopt renewable energy and recover heat or process water from production. As animal nutrition research sharpens focus on phosphorus utilization and sustainability, we invest in process upgrades and specialty co-product development. Nutraceutical and pharmaceutical markets want finer particle grades and confirmation of ever-lower impurity profiles. Government regulations on food chain safety and trace element exposures demand tighter documentation, more testing, and increased process transparency. Our own line improvements increasingly leverage process control software and inline analytics to minimize quality drift. All signs point to rising demand, particularly as emerging economies upgrade their food supply and healthcare infrastructure. Our role grows ever more critical—as reliable producer, as safety advocate, and as innovator supporting the next generation of phosphate chemistry.
People rarely think about the raw materials that support daily life, but as a chemical manufacturer, we see it constantly. Calcium hydrogen phosphate, sometimes called dicalcium phosphate, stands behind everything from the health of livestock to the tablets swallowed for dietary supplements. Chemists know the chemistry, but it’s the steady demand from agriculture, food, and pharma that proves its value.
The bulk of what we produce flows directly into animal feed. Cattle, poultry, swine—these animals need a steady source of both calcium and phosphorus for healthy bones, teeth, and egg production. Livestock producers count on feed formulas that deliver reliable nutrition, and calcium hydrogen phosphate gives them a safe, bioavailable source. This isn’t about niche applications; it’s the feed mills and farms around the world who trust this mineral every day because the animal’s health, and eventually the producer’s bottom line, hinges on consistent mineral content. Low-quality or contaminated feed supplements pose real risks, including decreased growth rates or metabolic problems.
You’ll spot this ingredient on food labels, too, not just in the vet’s office or the barn. Food producers turn to calcium hydrogen phosphate to fortify baked goods and cereals. The main reason is the reliable calcium boost, which helps address dietary deficiencies. It also acts as a leavening agent in flour mixes, breaking down under heat to release gases that help dough rise. In breakfast cereals, it goes into the premix, mixing right into the blend for consistent calcium in every bite.
Pharmaceutical companies face a challenge balancing good compressibility with the need for safe, inert fillers. Calcium hydrogen phosphate stands up to high-speed tablet presses, holding its shape without breaking down or acting up chemically. In vitamin tablets, both as a carrier and an active ingredient, this salt helps manufacturers achieve stable, solid pills that consumers can trust. Some of the largest multivitamin brands rely on our consistent particle size and purity to avoid production runs that jam machinery or cause failed tests.
Markets and end users expect reliability. That means controlling every step of the process, from the initial selection of raw calcium and phosphate rocks to the final stage of packaging. Small deviations in purity or trace contaminants spell trouble for product safety. We maintain detailed records, frequent batch testing, and transparency for downstream buyers. This matters for animal health, but it also protects children and adults taking supplements by mouth. On occasion, we see requests for custom specifications, such as extra-low heavy metal content or finer grades for infant formulas.
We hear concerns over sustainability and traceability. Sourcing cleaner raw materials or investing in safer processing keeps us a step ahead of regulatory changes and rising consumer awareness. Countries watching for imported feed materials or food additives don’t accept shortcuts. That pressure shapes our daily decisions, from investing in filtration to working with suppliers who care about sustainable mining.
Decades of feedback keep refining how we produce and what standards we hit. End users ask for clarity and reassurance—no surprises on their end. We see the difference every time shipments pass rigorous lab tests and livestock producers report improved growth or fewer health problems. In the end, success doesn’t hinge on a fancy formula, but on honest, thorough manufacturing and listening closely to what farms, food brands, and pharmaceutical companies need from us.
Calcium hydrogen phosphate shows up on many ingredient lists—breakfast cereals, bakery mixes, nutritional supplements, and more. We see questions about its safety come up from consumers, professionals, and regulatory bodies. As a producer, we have a front-row view of how it gets manufactured, tested, and used in foods. The raw materials come in as mined phosphate rock, and our quality team checks levels of trace elements. Each batch has to meet specifications for purity and heavy metals before it gets anywhere near a food facility.
Food-grade calcium hydrogen phosphate goes through strict review before manufacturers like us can even offer it to customers. Food safety authorities in the US, EU, China, and other large markets have included it on their approved additive lists. The US Food and Drug Administration (FDA) lists it as “generally recognized as safe” (GRAS) when used as an anti-caking agent, nutrient, or leavening agent in specified amounts. European food safety experts have reviewed available animal and human studies, looking at things like absorption, metabolism, and any toxicological data, as well as the body’s ability to handle extra dietary calcium and phosphate. Their decision: normal usage in foods does not present a risk to health.
Our engineers and chemists keep a close eye on the purity, particle size, and solubility profile in every batch. A key part of our job—making sure no excessive amounts go into end products. Too much added phosphate or calcium, over months or years, can affect kidney health for people who already have kidney disease. Food manufacturers typically use it at low concentrations, mostly to provide a calcium source or to control pH and texture. Both calcium and phosphate naturally occur in dairy, grains, meats, and many other foods. The body regulates their absorption tightly, and average intake from diet plus food additives stays well within safe levels for almost everyone.
All factory workers receive training on handling, storage, and personal protective gear. Finished material never leaves the facility without batch records, test results, and certificates of analysis. Once it hits a food production line, there's still batch traceability and documentation, plus the oversight of food safety inspectors. We see up close how much attention goes into keeping the ingredient clean, consistent, and free of contaminants like lead or arsenic. Well-run laboratories regularly test for these unwanted elements. Through this chain of controls, the risk of harm from calcium hydrogen phosphate in foods remains extremely low.
Consumers stay informed, and that’s a good thing. Any ingredient, even salt or vitamin C, can cause problems if grossly overused. Science and regulation work best when the manufacturing floor meets the lab and the law. As the science around food additives grows, we keep our procedures under review—always checking for updated safety data, changing international rules, and better ways to manage risk. Responsible production, regular monitoring, and honest dialogue keep this food-grade mineral safe for everyday use. From our experience, there’s no shortcut for vigilance and quality at every step.
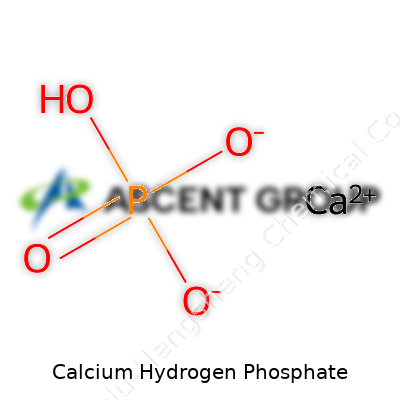
Working in chemical manufacturing connects people to raw materials in a straightforward way. Calcium hydrogen phosphate isn’t just another entry in a catalog. It’s a practical compound in daily production, and its formula—CaHPO4—shows the connection between laboratory principles and industrial-scale output. Labs sometimes call it dibasic calcium phosphate, and its composition reflects a particular chemical structure rooted in calcium, hydrogen, phosphorus, and oxygen.
For those of us who actually synthesize CaHPO4, the formula isn’t just a label. It points to a balance of ions in the finished product. One calcium ion, one hydrogen phosphate ion—it’s a mix that brings stability in storage. There’s no magic to it, just a lot of precise control over reaction conditions and purity checks. If a process used impure feedstock or ignored pH and temperature guidelines, the product might turn out as something else: monocalcium phosphate, for instance, or tricalcium phosphate. Such mistakes lead to visible issues in the plant and costly wasted batches.
Whether it ends up in animal feed, farming, or food-grade applications, calcium hydrogen phosphate has to meet strict standards. Calcium content comes from the kind of limestone or calcium carbonate used. The hydrogen phosphate fraction links to phosphoric acid feed. If those inputs slip out of the right ratio, or if water content climbs, the risk of off-grade batches rises. Many users don’t see how close quality control has to track the manufacturing run. Analytical chemists check every batch, and anyone cutting corners on this compound will show it in chalky, inconsistent product.
The name “calcium hydrogen phosphate” sometimes creates confusion. Some call it dibasic calcium phosphate; some refer to it by its mineral form, brushite or monetite, depending on crystal structure and water content. One process shift—like an extra dehydration step—can steer output from dihydrate CaHPO4·2H2O to anhydrous CaHPO4. These small tweaks have a big impact on downstream applications. In food, for example, the wrong calcium phosphate can mess up leavening, texture, or product label accuracy. In animal feed, small errors can trigger regulatory headaches.
As a manufacturer, we see daily the effect of these distinctions. Customers often come with questions after finding discrepancies in literature, or after a supplier drops documentation filled with ambiguous terms. That’s why direct communication about formula, hydration state, and purity always provides more reassurance than marketing copy. Product batches analyzed with modern techniques—X-ray diffraction for phase confirmation, inductively coupled plasma for elemental content—build trust in the supply chain.
So CaHPO4 is more than just letters and numbers. Consistent quality comes from discipline and transparency during production, not from clever branding or third-party repackaging. People rely on manufacturers who stand by their process and stay open about the exact composition they deliver. As formulas anchor our work, so does accountability for what leaves our gates and travels into finished goods around the world.
Anyone who's spent time producing Calcium Hydrogen Phosphate in tonnages knows the stuff seems pretty simple at first glance. Powdery, faintly granular, bland and stable. But behind that unassuming appearance, we’ve seen plenty of trouble over the years—caked bags, corroded machinery, surprising clumps that make precise dosing a chore. Storage isn't just a matter of stacking bags in a warehouse. What some call routine, we call essential for the integrity of the product and the people handling it.
Time and again, the labs and operations teams have flagged one culprit that throws off our Calcium Hydrogen Phosphate: humidity. The material pulls in water from the air. The moment a sack is left open in a damp corner, the powder starts clumping, sometimes forming a lumpy, brick-hard mess nobody wants to shovel. We know from experience and from the scraps of chemical theory that dihydrates can give up or take on water, shifting their composition if conditions swing too far in one direction. That’s not good in food applications, nor in feed, nor for phosphate processing.
Effective storage in our plants keeps moisture away at every step. Dehumidified rooms work best but aren’t always practical in bulk warehouses. High-quality liners—thick plastic, no pinholes—have become the norm for bigger volumes. For every batch, we double-seal bags and train warehouse teams to close loose sacks right away. Even in transit, we choose covered trucks or containers and avoid loading in rain.
Dust from other products can get into open Calcium Hydrogen Phosphate sacks. We’ve run into this in mixed-product warehouses. If urea, salt, or trace minerals make their way into our product, it’s no longer pure. One contaminated batch can ruin a week’s work if it goes unnoticed. That’s why most chemical manufacturing outfits—us included—store Calcium Hydrogen Phosphate away from volatile chemicals, acids, and fertilizers. Common sense says not to stack it where there’s potential for spillover or direct contact.
While Calcium Hydrogen Phosphate handles heat better than some raw materials, it hates extreme swings. High storage temperatures might not make it dangerous, but they can mess with consistency and shelf life. A warm, dry, shaded warehouse—protected from direct sun and far from boilers or hot pipes—has served us well. In colder regions, preventing condensation on packaging as the weather shifts ranks nearly as high as keeping rain out. Rapid temperature changes mean more condensation, more water intruding, and a higher risk for caking.
Crushed bags aren’t just an eyesore—they invite moisture and dust, and they frustrate everyone down the supply chain. Forklift drivers know to check load limits and keep bags off floors that pick up puddles or leaks from doors. Wooden pallets, though simple, form the backbone of good storage. Raising the product keeps it dry, and maintaining space between pallets allows for airflow so stray moisture can’t hang around and build up over time.
The best storage practices come from those working closest to the material. A seasoned warehouse hand will spot a ripped bag before it becomes an issue and will place new stock so older supplies move out first. Regular rotation reduces long stays in storage, which means fewer surprises down the road. Everything hinges on people paying attention, not just checklists or policy sheets.
After years of manufacture, it's clear that Calcium Hydrogen Phosphate rewards diligence with trouble-free storage and headaches for those who cut corners. If the storage is done right, the product stays free-flowing and steady, ready for whatever the next step demands. It might sound like simple discipline, but it’s what allows us to deliver material we can stand behind, batch after batch.
In our manufacturing experience, calcium hydrogen phosphate turns up in more processes and products than many realize. It provides calcium and phosphate, both essential for the body and widely recognized by food authorities around the world. Many baked goods, cereals, supplements, and even animal feeds make use of its mineral value. Still, familiarity with a material like this creates an obligation: acknowledging and managing any risks that come with it.
Most people tolerate calcium hydrogen phosphate in routine concentrations. Our plant teams have fielded questions over the years about digestive discomfort. The truth is, overconsumption of most calcium compounds runs the risk of minor bloating, constipation, or gas. These side effects often show up in people already consuming diets rich in calcium through dairy and supplements. In rare cases, especially in people with pre-existing kidney challenges or parathyroid issues, high dietary phosphate and calcium can contribute to imbalances. Excess calcium can increase the chance of kidney stones. Anyone with a history of these conditions needs to handle all calcium-rich products, supplements, and additives with more care.
We pour a lot of effort into ensuring raw materials stay pure, but questions about allergies are reasonable. Calcium hydrogen phosphate itself does not belong to the class of allergens like nuts, gluten, or soy. The focus in our quality control lab rests on preventing cross-contamination and verifying source purity. Occasionally, impurities from upstream raw mineral sources become a concern, especially with heavy metals. As a manufacturer, we stick to routine heavy metal analysis, batch testing, and documentation. Regulations encourage these routines, but industry credibility depends on exceeding standards.
Some customers ask about the risk of long-term exposure. For employees, handling isn’t an issue when safety measures are followed. In the broader public, the largest risk connects to high total intake of calcium and phosphate, not the compound itself. Too much phosphate over time has drawn some scrutiny in nutrition science, especially with processed food as a growing part of global diets. Elevated phosphate can affect cardiovascular and bone health in at-risk populations: elderly, those with chronic kidney concerns, and those taking certain prescription drugs.
Many people confuse calcium hydrogen phosphate with other forms of phosphate or with calcium carbonate used as antacids. Each material brings its own benefits and quirks. Honest labeling, clear communication with downstream users, and close partnerships with nutritionists and product developers help prevent misuse and overexposure. Our teams also find that collaborating on fortified foods and supplements works best with tailored dosage instructions and a willingness to answer regulatory inquiries directly.
A manufacturer’s responsibility doesn’t end at the gate. We help customers educate themselves, improve transparency, and address possible overages or label errors when they arise. New research, updated regulations, and customer feedback guide updates in our practices. As the maker, we see it all starts with vigilance and a long-term view on health and safety.