Copper acetate monohydrate traces its roots deep into the history of both chemistry and manufacturing. Early alchemists and apothecaries recognized copper salts' vivid blue and green hues, long before chemical formulas entered daily industrial life. Copper acetate, in particular, evolved from medieval practices involving verdigris, which artisans scraped from weathered copper for use as both pigment and a mordant. Modern synthesis began to take off with systematic studies on copper compounds in the eighteenth and nineteenth centuries. Advances in analytical chemistry helped define copper acetate’s precise composition, leading manufacturers like us to refine our methods—bringing reliable, high-purity products to customers worldwide. These improvements didn’t happen overnight. Instead, they reflect generations of persistent work, continual troubleshooting, and direct feedback from application laboratories. The accumulated lessons shape how we approach raw materials, quality control, and innovation today.
Copper acetate monohydrate stands out among copper salts because of its solubility, stability, and reactivity. In the chemical manufacturing community, this is a backbone material for organic synthesis, catalyst development, fungicide formulation, pigment production, and more. Few copper compounds deliver this combination of consistency and versatility. As a crystalline salt, the monohydrate form offers improved handling characteristics and longer shelf life over the anhydrous form, supporting smoother integration into chemical processes. Most chemists who rely on it appreciate its straightforward behavior in both lab-scale trials and commercial-scale batches. It serves as a trusted intermediate, hopping between glassware, reactors, and downstream processes, so robust production protocols matter to everyone depending on it.
Copper acetate monohydrate appears as blue-green crystals, easily recognizable by anyone who’s spent time around copper chemistry. Our experience handling this material reinforces the need for strict water content control, since drier conditions yield a powdery anhydrous version that behaves differently and causes processing headaches. The compound dissolves readily in water and various alcohols, a trait essential for consistent dosing in reaction systems. Chemically, it possesses moderate oxidizing properties, and its copper (II) center interacts readily with ligands and reactive organics. From a manufacturer’s perspective, subtle changes in particle size or hydration state can ripple through batches—driving us to monitor every step between starting ingredients, crystallizer operation, drying regimes, and final packaging.
Specifications for copper acetate monohydrate don’t just keep regulators happy; they serve as the backbone for user confidence and operational safety. We base our technical sheets on decades of standardized tests, including purity (typically above 98 percent for technical grades), water content, insoluble matter, and trace metals. Accurate labeling means more than a legal requirement; it communicates real, actionable knowledge to research labs and process technicians who demand reproducibility. Inside our factory, every drum and bag carries unique batch identifiers, hazard classifications, and storage guidelines. Our priority is not to pile up paperwork, but to make the relevant information available for anyone—from a junior chemist up to a plant manager—who needs to judge material suitability on the spot.
Making copper acetate monohydrate is a story of chemistry and equipment. Direct reaction between copper (II) oxide or copper carbonate and acetic acid allows us to fine-tune the conditions for high yield and purity. Reaction temperature, acid concentration, and agitation must be managed closely since any shortcut can introduce impurities or uncontrolled particle formation. Excess acetic acid is strictly avoided to limit unwanted by-products and corrosive risks further along the process line. Crystallization doesn’t just separate product from solution; it also defines the lot’s particle size and water of hydration. In our experience, controlling cooling rates and drying under low heat prevent partial dehydration—a detail sometimes missed by less experienced operators. We continually invest in automation and sensor technologies, letting us track pH trends, conductivity, and crystal morphology. These deep process controls lower risks of batch failures and support customers with the most demanding applications, whether they need kilo, tonne, or container-scale quantities.
Copper acetate monohydrate reacts as a Lewis acid and participates in redox steps. These features open up a range of synthetic paths for manufacturers and research users alike. Acetate groups can be displaced by stronger ligands, creating new copper complexes for catalysis or electronics. When heated or mixed with certain organics, it becomes a useful oxidant for transforming hydrocarbon substrates, fostering cross-coupling reactions, and generating value-added intermediates. In surface chemistry, its reactivity underpins the creation of nanostructured copper oxides—materials increasingly relevant for electronics and green energy sectors. Our R&D team often works alongside industry partners to refine these modifications, testing how altered stoichiometry, solvent systems, or reaction kinetics change the final outcome. We know from experience that even minor tweaks to process, solvent, or starting copper source can have far-reaching effects on downstream performance.
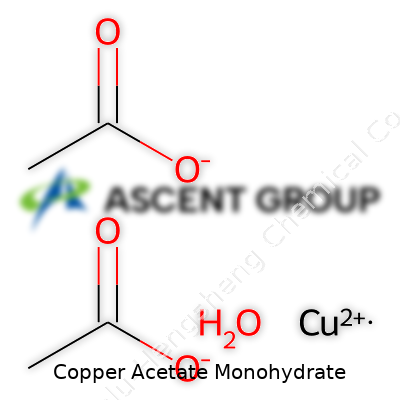
Those who buy or use copper acetate monohydrate understand it appears under several synonyms, including cupric acetate monohydrate and blue verdigris. These names crop up in academic papers, regulatory filings, and old technical manuals. The chemical formula, Cu(CH3COO)2·H2O, provides precision across markets, yet end users often navigate a tangle of local conventions and product grades. Direct communication clears confusion; as manufacturers, we align our documentation and SAP systems with common nomenclature and keep librarians, purchasing staff, and project leads on the same page. A shared vocabulary prevents costly mistakes and supports the wider industry’s march towards quality and compliance.
Copper acetate monohydrate demands respect both in bulk storage and routine handling. We see the risks—eye and skin irritation, inhalation hazards, reactivity with incompatible materials—and train our workforce thoroughly. Well-ventilated facilities, personal protective gear, spill management drills, and clear signage shape daily operations on our shop floor. Waste streams, spent solutions, and air emissions are treated in compliance with environmental guidelines, reflecting the trust placed in us as direct stewards of chemical safety. Our internal incident reviews drive improvements, and we routinely consult safety data sheets, published research, and regulatory bulletins to update our policies. These measures protect our teams and community at large, not just from fines but from real-world harm.
Copper acetate monohydrate serves a diverse base—catalysis, electroplating, dye and pigment manufacture, antimicrobial coatings, wood preservation, and laboratory synthesis, to name a few. In catalysis, it helps drive C–C and C–O coupling reactions with greater selectivity and speed. In biochemistry, its precise copper content assists with enzyme studies and protein structure mapping. Our biggest customers often come from the pigments and wood treatment sectors—seeking copper’s protective qualities and hybrid color chemistry. We also notice a surge in specialized electronics users, where it fits into conductive ink prototypes and battery precursor trials. Scaling up from grams to tonnes gives us unique production insights; each segment pushes us to refine processes, eliminate contaminants, and respond to new technical challenges raised by customers refining their products alongside ours.
R&D for copper acetate monohydrate never stands still. Our chemists work with academic and industrial collaborators to tune properties for specific needs—launching new grades with tweaked solubility, particle sizes, or impurity profiles. Laboratory-scale synthesis often exposes subtle effects that larger plants must address: thermal stability, ease of filtration, or by-product management. We channel feedback into next-generation reactors, improved analytical tools, and custom packaging for sensitive end uses. Technological trends, like movement towards greener synthesis or waste minimization, challenge manufacturers to balance tradition with invention. We keep detailed records, run inter-laboratory comparisons, and invite peer review of our protocols, maintaining scientific rigor every step from material prep to final shipment.
Copper acetate monohydrate’s safety profile stems from both copper toxicity and acetate’s mild corrosiveness. Extensive literature highlights copper’s essential but potentially toxic biological role, especially at high exposures. We track permissible exposure limits mandated by agencies such as OSHA, REACH, and the ECHA, and conduct regular operator health monitoring. Our responsibility goes beyond lab protocols; we design workplace rotation schedules, install fume hoods, and invest in spill containment systems to guard against acute and long-term risks. Data from in vitro and animal studies inform our product stewardship. Constant technology reviews and regulatory updates keep us ahead of changes that could affect labeling, permissible limits, and export restrictions. For downstream users, providing accurate MSDS and targeted education programs helps mitigate misuse and accidental exposure, closing the gap between theory and practice.
Copper acetate monohydrate’s future hinges on sustainability, advanced manufacturing, and new application development. Green chemistry pushes us toward waste minimization, solvent recovery, and renewable feedstocks, aiming for minimal environmental impact. Progress depends on finding cleaner production routes, recycling copper from electronic waste, and integrating process analytics for real-time control. Technical advances in electronics, antimicrobial coatings, precision agriculture, and battery research each create new demands for ultra-pure or custom-formulated materials. We see increasing interest in single-crystal forms and functionalized copper complexes, shaped by trends in nanotechnology and electronic device miniaturization. The ability to track the material’s journey—raw copper source to finished acetate—shapes customer trust, regulatory compliance, and our standing as a responsible manufacturer. This path forward requires continuous learning, investment, and collaborative partnerships, connecting old chemical wisdom with tomorrow’s opportunities.
Those of us working on the ground in chemical manufacturing know the daily challenges that come with producing specialty materials like copper acetate monohydrate. In our own operations, this compound has become a staple for applications that demand reliability and consistency. If you have ever worked in catalyst production, textile processing, or wood treatment, copper acetate monohydrate has probably found a place on your bill of materials. It’s not just another copper salt—its role cuts straight to the heart of several chemical processes that drive important sectors.
Chemical synthesis needs copper acetate monohydrate for reactions like the Wacker process, a widely used method for turning ethylene into acetaldehyde. This process is a vital link in the production of acetic acid and several solvents. Reliable catalyst function directly impacts cost, yield, and safety, and that's worth more than any abstract benefits. From our own observation, the purity of copper acetate monohydrate determines success or failure in these catalyst regimes.
Ask someone in textile mills or pigment houses about the color blue in their products, and copper acetate monohydrate will come up. The chemical reacts with various materials to form blue and green pigments, such as verdigris, that still find use in dyes, paints, and inks. Consistency in color comes down to dependable chemical supply—we handle that at scale with strict controls over particle size, water content, and purity. There’s no room for shortcuts if you want to avoid product recalls and unhappy customers.
In preservation, copper acetate monohydrate helps formulate fungicides and anti-mildew solutions for lumber yards and carpentry shops. Treated wood for outdoor use depends on copper’s fungicidal behavior. Blends with other copper salts extend a product’s lifespan, making it possible for fences, playground equipment, and outdoor furniture to resist decay. As a manufacturer, seeing our product integrated in this value chain puts a strong focus on environmental responsibility and traceability.
Chemistry classrooms and research labs rely on copper acetate monohydrate for crystal growth experiments or as a starting reagent for testing reaction mechanisms. As a direct supplier to universities and technical schools, our focus lands on producing small, highly characterized batches for reliable classroom results. Accuracy matters more than volume in these shipments, and transparency in raw material sourcing is non-negotiable.
As demand grows across these industries, pressure mounts on manufacturers to ensure responsible sourcing. We have responded by investing in closed-loop systems and greener synthesis methods that reclaim copper from waste and reduce energy loads. The chemical industry faces regular criticism for environmental impact; practical action—like improving waste management and reducing energy needs—separates talk from progress. Regulations are moving fast, so our future growth relies as much on compliance and transparency as technical expertise.
True value comes from understanding where and how copper acetate monohydrate enters the supply chain—then optimizing each link. Through tighter documentation, closer relationships with end users, and better control over purity, we have cut down on preventable failures and surprising product issues. After years in manufacturing, I can say that these practical details matter far more than buzzwords or hype. Industry needs compounds that do their job, every time, without fanfare or fuss. We believe copper acetate monohydrate delivers exactly that.
Manufacturing copper acetate monohydrate day in and day out brings one simple truth into sharp focus: every detail matters, including the chemical formula. Our team learns this in the plant, not just the lab. To the direct question—copper acetate monohydrate has the chemical formula Cu(CH3COO)2·H2O. The formula marks each molecule as holding one copper ion, two acetate groups, and one water molecule tightly bound in the crystal.
Why point out something so specific? Because process mistakes don’t stem from poor intent but from missing the smallest variable. The structure matters since the single water of hydration changes behavior compared to the anhydrous form. In storage, we see cakes of blue-green crystals absorbing moisture from the air, not powdering down like anhydrous copper acetate. The stability and physical handling differ. Down the line in formulation work—say in pigment manufacture, textile dyeing, or even as a catalyst precursor—process consistency hinges on the formula. Miss the hydrated water, and batch yields can drop, reaction conditions skew, or final applications present inconsistencies.
Precision around the chemical formula doesn’t just save money. It keeps people safe. Copper compounds share a set of hazards; the monohydrate, with its recognizable formula, gets matched with the correct procedures in labeling, storage, and disposal. Regulatory agencies call for proper reporting, transparent documentation, and traceability. A missed water molecule in the chemical formula leads to wrong entries and headaches during audits, but it can spark far greater problems in case of an emergency. From our shop floor to paperwork for customs, it’s the written Cu(CH3COO)2·H2O that legally and chemically ties it all together.
Production isn’t always a textbook operation, either. During synthesis, copper(II) oxide or basic carbonate gets thoroughly reacted with acetic acid; the process yields copper(II) acetate and liberates water precisely because the monohydrate form will crystallize under those conditions. We continually monitor the product to confirm the expected hydration—not too much, not too little. X-ray diffraction and Karl Fischer titration replace any guesswork. Cutting corners or skipping checks courts disaster: excess water alters solubility and handling, which can cause entire batches to divert to waste instead of heading to customers.
As a supplier, every end user expects what’s on the label matches what they receive. That’s not just a matter of trust. Analytical chemists and process engineers rely on clean, accurate formulas so their reaction stoichiometry balances, and byproducts don’t take them by surprise. Toxicity studies and environmental impact assessments ride on having identifiers like Cu(CH3COO)2·H2O, not a guess or a vague description.
As demand rises for greener, lower-impact chemicals, the stakes increase for precise, declared formulas. Manufacturing has shifted from broad-brush approaches to well-monitored batch and continuous processes, and the chemical name now gets a microscope’s attention. No mix-up or shortcut pays off; strict attention to detail always beats vague description. Those who work with copper acetate monohydrate appreciate how a small chemical formula means a lot beyond the page—affecting practical operations, compliance, worker safety, and trust at every stage.
Years spent producing copper acetate monohydrate have shown that safe storage isn’t just a checkbox for compliance, but a daily priority that protects both quality and safety. This powder absorbs water readily and reacts with the air around it, resulting in unwanted caking and sometimes degraded material performance. Letting it sit in a humid or variable environment brings complaints about inconsistent color in downstream processes or reduced solubility. Even small changes in storage conditions bring headaches—so we design our facilities around what this chemical demands.
Copper acetate monohydrate’s affinity for absorbing atmospheric moisture means dry, stable air is more valuable than any fancy labeling or packaging. In our plant, we choose well-sealed, tightly lidded containers made from high-density polyethylene, lined metal drums, or glass. Leaving the powder exposed, even for an hour, creates clumps that complicate both dosing and dissolution later. There’s no shortcut: A climate-controlled warehouse, with relative humidity below 50%, minimizes these issues. Exposing the chemical to frequent temperature swings triggers condensation inside containers—this, in turn, triggers complaints about material sticking to the sides and erratic batching.
Reliable storage means more than just keeping moisture out. Any manufacturer who’s dealt with clients in pigments, catalysts, or school laboratories knows high purity must be preserved up to point of use. We started reserving dedicated storage areas, away from strong oxidizers, acids, or volatile solvents. Any cross-contamination quickly alters chemical properties or—worse—leads to reports of unpredictable side-reactions in the customer’s process. We train every warehouse worker to recognize the signs: Watch for leaks, powdered spills, or discoloration on the packages. Fast action stops small issues growing larger.
Whether shipping in 5-kilogram laboratory packs or full pallets for industry, we select packaging to match storage environment and delivery timing. For customers planning to open and reseal frequently, triple-sealed liner bags offer protection for months. Those storing bulk product for longer periods get drum liners plus inner bags, each tied with moisture-indicating cards. Every extra layer comes from past mishaps—a single summer storm or warehouse leak can lead to a whole shipment lost to degradation.
In our own experience, even experienced staff sometimes miss packaging flaws or early moisture damage. We hold regular reviews, walk warehouse lines, and encourage open reporting. New storage protocols or climate alerts go out promptly, so everyone acts before conditions turn costly. Having routine checks for both ambient humidity and visual package inspections keeps us ahead of surprises.
Real-life manufacturing experience confirms that storing copper acetate monohydrate is about more than loss prevention. It’s about delivering consistent, high-quality material with predictable behavior. Every choice in how to store, package, and monitor this compound draws directly from hard-earned lessons and ongoing vigilance. These habits shape trust with partners, researchers, and industries relying on dependable chemicals.
Working on the manufacturing floor, you start to develop a healthy respect for the materials you handle, and copper acetate monohydrate doesn’t let anyone get careless. It wears its bright blue color like a warning. Our teams always follow established practices – not because the paperwork says so, but because they’ve witnessed the results of inattention. Skin contact can leave irritation. Inhaling its dust leads to a nagging cough, sometimes nausea. Eyes smart quickly. That’s not rumor – that’s from daily life in any facility refining or blending copper acetate for shipment.
Copper compounds won’t sneak up with exotic risks, but their toxicity deserves respect. Copper ions, once absorbed, build up in body tissues. Acute overexposure starts with stomach pain and vomiting; long-term routines with poor controls may eventually stress the liver or kidneys. Most globally accepted workplace limits, like OSHA’s, set exposure caps for copper dust and mists – not out of regulatory habit, but learning from real cases. Copper acetate doesn’t transform into something completely unexpected; it acts like other copper salts, except maybe a touch faster because it dissolves so well in water and enters the body more readily.
Nobody leaves open bags or containers sitting around. Humidity causes copper acetate monohydrate to cake up, and airborne dust soon finds its way around the room. A good ventilation system isn’t “optional” – it keeps air quality within limits. Handling never means bare skin. Gloves and splash goggles make for better shifts and fewer trips to the eye wash. We control storage, keep it away from acids or strong bases. Labels run clear and simple: not just for meeting rules, but so anyone can follow the thread in a noisy, busy work room. Everyone knows, from the shift supervisor to maintenance, how to handle a spill. We keep copper compounds well clear of food areas, avoid cross-contaminating with other raw materials, and collect all waste with the same respect we give to higher-profile chemicals.
Copper acetate monohydrate ends up in many industries, from catalysts to fungicides. The concerns rarely stop at the plant gate. Plants that forget careful disposal may see increased copper levels in local waterways. Copper builds up fast in aquatic systems. Even a little over time stresses fish and other organisms, altering the entire ecosystem. Fines and warnings from regulators encourage compliance, but the strongest learning comes from local community reactions. Once a river or pond shows signs of copper buildup, the trust of neighbors takes a long time to recover.
Nobody in the manufacturing world can afford to treat copper acetate monohydrate lightly, nor fall back on checked boxes. Training sticks best when workers recall watching a careless peer suffer skin burns or seeing blue stains appear on hands without proper barriers. Sharing lessons keeps teams tighter and mistakes fewer. Engineering improvements – closed conveyors, dust collection, and sealed packaging – cost money, but offset longer-term worries about injury or environmental damage. Professionals turn knowledge into habit, so the hazards stay controlled, the team stays healthy, and local water stays clear.
Out on the production line, purity is a number that holds real meaning. As a chemical manufacturer making copper acetate monohydrate, we measure purity every shift. Clients rely on reliable copper content for research and manufacturing, whether they’re in catalysis, pigments, or agrichemical sectors. Delivering a product at or above 98 percent purity isn’t just a figure—it’s a reflection of our commitment to good chemistry and respect for the work our customers do with our product.
The outcome depends on what you start with. Every drum of copper metal and batch of glacial acetic acid we unload is traced, with certificates checked and independently matched against our own assays. Variability in copper quality affects downstream purity. Sourcing metal grades with trace impurity control forms the first safeguard, not just in paperwork, but in real-world filtration and melting steps.
We see surprises on the plant floor whenever controls are treated as an afterthought. Analytical staff conduct spot checks with titrations and spectroscopy—not merely to fulfill a spec sheet, but to understand the chemistry at play. Some labs look for shortcuts. We pull random samples and run copper content and residual analysis even if the process has never missed the mark for months. This approach prevents drift and helps us catch issues before they multiply.
Humidity, temperature, and air quality inside the reactor room are not just details logged for compliance; shifts in these conditions nudge a batch outside tight tolerances if overlooked. Introducing even trace amounts of unwanted ions can change the color, solubility, and ultimately, the reliable function of copper acetate monohydrate. So, operators work inside clean environments, with every vent scrubbed regularly, and water of controlled purity.
Purity level needs to mean what labs and process engineers expect. Unintended metallic residues or unreacted organics from slapdash production carry through to downstream operations. We stay in touch with users who run the acetate in polymer modification, analytical chemistry, and surface treatment. If a lab calls in, worrying about unexpected reactivity or trace contamination, we keep records and invite customers to review analytical results. This dialogue helps us fine-tune approaches so the product matches performance expectations each time.
Over the last years, tighter quality audits and higher stakes have become the norm. New international standards push for more precise limits on iron, lead, and other contaminants. We invested in higher-resolution analytical equipment and keep a tight feedback loop with our raw material providers. Questions about source mines, production conditions, and even packaging choices surface regularly: plastic drums vs. lined steel containers—each has implications for purity over time. Our move to moisture-protected packaging came after hearing customer concerns around caking and hydrolysis, especially for long-distance export.
Sometimes, price pressures creep up, usually sparked by swings in the cost of copper or shipping. Opting for cheaper raw ingredients can be tempting in the short term, but the risk to purity remains too high. Losing trust through unpredictable performance would cost far more than any savings upfront. In our experience, open conversations with partners around cost, technical requirements, and regulatory changes go farther than top-down directives. These discussions lead to real improvements in both purity and how dependable our product stays year after year.