Dimethylformamide (DMF) has shaped the course of the specialty and bulk chemical industry ever since its initial synthesis in the mid-20th century. Reflecting back, plants like ours recognized early how DMF changes the game in solvent technology for polymers, pharmaceuticals, and agrochemicals. Rapid expansion in synthetic fibers, resins, and drug development fueled demand, turning DMF from a niche lab material into an essential ingredient across continents. As a producer, we witness demand that traces the rise of advanced coatings and high-performance fibers, the spread of polyurethane processes, the drive for higher yields in organic synthesis, and now, the needs of modern battery chemistry. From the viewpoint of operations, DMF isn’t some relic of yesterday’s R&D—it continues to anchor chemical transformation on large and small scales.
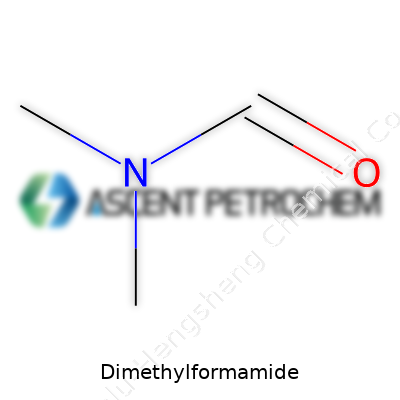
DMF isn’t a one-trick solvent. With its molecular structure—an amide where two methyl groups flank a formyl group—DMF brings a set of performance characteristics few other liquids match. Its clarity, near odorless profile, and full miscibility with water, alcohols, and common chlorinated and aromatic solvents make it valuable for dissolving polar and non-polar compounds. Over years of batch production, the value lies in its polarity: DMF dissolves high-molecular-weight polymers (like polyacrylonitrile and PVC), supports acrylamide and peptide synthesis, and acts as a reaction medium for organic halides and esters. A boiling point just under 153°C, a flash point near 58°C, and low volatility have impacts every day on our production design, handling equipment, and process safety. For those using DMF, these numbers don’t stay on paper—they drive workflows on the shop floor.
For anyone drawing off storage tanks in a chemical plant, consistency isn’t optional—every shipment of DMF must hit strict technical specs. Purity standards remain above 99.8 percent for most critical uses. Moisture content rarely exceeds a few hundred ppm, given the high dehydration standards, since excess water can ruin sensitive reactions. Acid value stays minimal to avoid catalyst poisoning in specialty polymer work. Over the years, shifts in regulation brought sharper focus on clear labelling, hazard statements on containers, and traceability right down to the batch level, especially because of DMF’s safety classification. As a manufacturer, regulatory and client audits never take a break—labels bear UN numbers, controlled substance warnings for certain countries, and GHS pictograms codified by ever-tougher global standards.
Industrial processes for making DMF have evolved with both demand and regulation. Today, our core method couples dimethylamine with carbon monoxide under pressure, typically using methanol as a formylating agent and a copper or alkali catalyst. Safety engineering and process optimization reduce byproduct formation and help us recover unreacted amines for re-use. Maintaining the right reaction temperature and continuous separation of the aqueous and organic phases have meant fewer shutdowns. In the early days, alternative routes tried to start from formic acid derivates, but high yields and reliable quality made the methylamine-carbon monoxide process dominant. Waste streams, especially containing amines and carbonyl residues, require careful onsite treatment before effluent discharge—compliance hinges on maintaining in-process and end-of-pipe control, highlighting operational discipline as critical as technical prowess.
Organic chemists lean on DMF for its role as both solvent and reagent. The formamide group in DMF can release dimethylamine under strong conditions, opening options in alkylation or transamidation chemistry. The Vilsmeier–Haack reaction leverages DMF’s structure—mixing with phosphorus oxychloride, DMF becomes a powerful formylating agent for aromatic aldehyde synthesis. On plant scales, these reactions aren’t just academic: our process safety controls must manage exotherms and toxic off-gassing during these modifications. In nucleophilic substitution chemistry, DMF not only supports the reaction but can itself get involved through “dimethylaminomethylation” in some routes. As regulations scrutinize byproduct formation, we shift R&D into pathways that curb unwanted DMF decomposition, minimize N,N-dimethylamine, and ensure recoverability from spent reaction mixtures.
Across documentation and global trade, DMF appears under several names: N,N-dimethylformamide, formic acid dimethylamide, and just “DMF.” Some legacy markets also refer to it by German or Russian trade names. For us as a direct producer, getting this nomenclature correct avoids confusion and shipping errors. SDS documentation, customs declarations, and GHS-labelling must reflect each jurisdiction’s language and regulatory coding. The same compound, but country-specific synonyms, demand vigilance throughout the supply chain.
No serious manufacturer ever treats DMF lightly. It penetrates skin and membranes, and inhalation brings acute and chronic toxicity risks, mainly liver and reproductive toxicity concerns. As stewards of plant safety, our teams enforce closed processing—automated filling systems, vapor recovery hoods, and stringent PPE for every operator. Occupational exposure is tracked via biological monitoring; periodic air sampling in transfer rooms tells us if something deserves a fix. Over years, regulatory tightening—from OSHA in the US to European REACH protocols—forces us to stay ahead on engineering controls and hands-on staff training. Each incident, whether splash or leak, is investigated in detail, with immediate lessons transferred into operational protocols.
The profiles of industries consuming DMF show how indispensable it remains. Polyacrylonitrile fiber, the precursor for carbon fiber, owes much of its processability to DMF’s solvent performance. In crop protection, DMF dissolves stubborn active ingredients, supporting formulation of advanced herbicides and fungicides. Pharma R&D teams reach for DMF to speed up peptide coupling and improve solubility in lead compound screening. Production of synthetic leathers, adhesives, and paints wouldn’t meet style or durability standards without DMF driving resin solubilization. The lithium battery sector, especially as energy storage shifts toward high-performance separators, finds DMF essential for making polar polymer membranes. Our manufacturing teams track shifts and spikes in these markets directly from quarterly order volumes and the technical queries that land with each PO.
Few chemicals see as much continuous R&D scrutiny as DMF. Research pushes boundaries on improved recovery systems to minimize environmental loss and develop catalytic routes that avoid hazardous intermediates. Our R&D colleagues closely watch literature on less toxic alternatives (like dimethyl sulfoxide or N-methylpyrrolidone), but DMF’s price-performance ratio means no clear winner has emerged for many applications. Over the past decade, green chemistry initiatives prompted continuous upgrading of our plant: thermal oxidizers handle fugitive vapor, and closed-loop reclamation systems squeeze out extra recoveries from what would have been process waste. Partnerships with downstream users—pharmaceutical API plants, fiber spinners, membrane manufacturers—result in collaborative projects to recycle DMF from spent solutions, cutting costs and solid waste at both ends.
Toxicology teams and regulators assign DMF a profile that cannot be ignored. Chronic exposure studies point clearly to liver stress and changes in reproductive health markers in both animals and exposed workers. We’ve sponsored and participated in longitudinal health studies, tracking employees working directly with DMF for decades. Results from these studies build the backbone of our internal risk assessments: daily and shift-long airborne concentrations must remain under cited occupational limits, while biological monitoring provides a safety net against undetected cumulative exposures. Wastewater, plant residue, and tank-cleaning effluents must all meet local and international frameworks before their safe release. We engage regularly with environmental health researchers to track emerging findings—and adjust controls as new data or regulatory targets surface.
Future prospects for DMF turn on two competing forces. On one hand, new technologies—advanced batteries, specialty polymers, engineered resins—are driving net growth in DMF usage, especially in Asia and growing economies. On the other, rising regulatory headwinds over worker health and environmental impact push customers and ourselves to consider alternatives, invest in abatement, or overhaul production cycles. For every plant expansion project, we invest heavily in vapor recovery, zero-discharge effluent systems, and operator training. Collaborative industry efforts seek both better analytical methods to detect low-concentration DMF in air and water and alternative solvent chemistries that don’t compromise performance or price. As process chemists and plant managers, we see every day how DMF bridges legacy materials science with modern industrial demands—and shapes both challenges and opportunities right out on the factory floor.
At our facility, we see DMF get called off the tanks every day for a hundred different reasons. This compound keeps production lines moving in pharmaceuticals, agrochemicals, fibers, and electronics. DMF gives chemists room to maneuver, thanks to its ability to dissolve everything from polymers to salts. Factories rely on that solvency when they’re chasing high yields and pure product. For many colleagues on the floor, that translates to tighter process control and fewer reworks down the line.
Some people judge a solvent by its smell or color. We look at the reaction flask. DMF’s polarity and low evaporation nature let it stick around during long syntheses. This turns what should be a temperamental reaction into something consistent. That saves time repairing vessels or flushing lines clogged with residues. We’ve run experiments with other polar solvents, and few match DMF’s ability to keep batch runs steady through temperature swings.
Acrylic fiber plants keep a steady stream of DMF to their spinning machines. It dissolves the raw acrylonitrile polymers and forms the kind of dope that flows through spinnerets to make the soft fibers found in sweaters and fleece. Fewer blockages mean higher uptime and less frustration. Over in electronics, DMF works wonders in cleaning and degreasing printed circuit boards. Its ability to remove stubborn organic contaminants shortens downtime between product changeovers, a real asset to teams racing against delivery deadlines.
We don’t compromise with the active pharmaceutical ingredient teams. They rely on DMF to dissolve a wide range of intermediates. It’s not just about getting chemicals into solution but helping them react at the scale drug companies run. Process robustness directly affects people’s health. When customers ask how we enforce purity, every operator on this shift knows the column runs, the distillations, and the GMP logs behind each drum. It’s a commitment we keep, because shortcuts show up fast in medicine, and the consequences aren’t abstract—they’re personal.
No one in manufacturing shrugs off DMF’s hazards. Vapor controls and closed-circuit transfer keep our guys safe, and we audit those systems daily. Understanding the health impacts pushes us to keep exposures in check and invest in better detection. Industry-wide, there’s rising interest in alternatives, but substitutes bring their own issues—lower dissolving power, higher costs, or retooling equipment. Our experience says total replacement remains a tough ask, but smarter use, including solvent recovery and recycling, does cut waste and risk. Our solvent distillation columns run nearly round the clock, driving home the fact that responsible DMF handling is less about paper policy and more about discipline on each shift.
DMF production carries real responsibility. We see governments tightening workplace and emissions regulations, and we meet those with honest reporting and investments in environmental controls. On the frontlines, our teams learn to respect DMF’s strengths and be frank about its limits. Down on the shop floor, we don’t see this as just another solvent—it’s a linchpin in innovation, shaping products that land in homes, clinics, and factories around the world.
Every day, production teams watch tons of dimethylformamide flow through reaction tanks, mixers, and storage lines. With a clear liquid like DMF, hazards don't announce themselves with fire or smoke. Workers see little more than a mild scent and a slick surface under their boots. DMF’s threat hides in its easy absorption through the skin and lungs.
Decades operating reactor systems and cleaning up spills have taught us that direct contact brings risk. Workers absorbing DMF often see mild skin irritation at first, but repeated or long contact causes dermatitis and even liver effects over time. Breathing in vapors over a shift may bring headaches, nausea, or signs of nervous system disruption. Routine medical exams sometimes flag irregular liver function in operations teams exposed for long periods. These effects do not suddenly appear overnight, and for years, some plants ran with no one thinking much about a little skin splash or a whiff from an open drum. Now, the evidence piles up in health studies—chronic exposure raises risks much more than most used to believe.
Experience shows risk management works best on the factory floor. At our production site, operators wear gloves and aprons by habit. Ventilation systems pull away vapors from open process tanks, and closed transfer systems keep DMF inside the pipes. Still, people make mistakes. A small spill during drum changeovers or a failed goggle seal leaves more exposure than anyone wants. Over the years, monitoring results painted a clear picture: even modern exhaust fans leave a workplace with trace amounts in the air, unless crews stay sharp and systems run hot all shift.
Researchers link dimethylformamide to liver disease from chronic exposures, and regulatory agencies set limits for workplace concentration. In Europe, legal exposure levels dropped in recent years after new reports of harm, driving plant managers to upgrade exhaust and leak detection. In our experience, maintaining DMF concentrations below official guidance marks means checking more than just average readings—peak short-term exposure still happens at batch dump or filter cleaning stages. Quick handling and attention to small leaks go further in stopping trouble than paper rules alone.
Some production runs can swap DMF for less hazardous solvents, but not every process adapts. Polyurethane manufacturing, acrylic fiber spinning, and electronic chemicals still run best with DMF. At our facility, the low boiling point lets teams recover and recycle much of it, but the recovery units need airtight seals and real-time detection in the event of loss. Training new staff doesn’t stop at handing out gloves—hands-on safety drills and sharing stories of near-misses matter even more. After a shift, health checks and clear records help us spot problems before they grow.
Today, tighter rules and better science keep plant staff safer than in past decades. Still, safe work with DMF means more than following a book of rules: it’s built into every transfer, inspection, and repair in the plant. Safety culture takes honest reporting, smart response to alarms, and a focus on real conditions—factors only chemical manufacturers who live with DMF every day can truly grasp.
The chemical formula for dimethylformamide reads as C3H7NO. To us in manufacturing, these five symbols translate into a solvent that drives countless industries. Each batch leaves our reactors with a story behind it: ensuring the right mix of carbon, hydrogen, nitrogen, and oxygen—three carbons, seven hydrogens, one nitrogen, and a lone oxygen—forming a molecule that continues to earn its place in labs and production floors worldwide.
We have watched this formula support transformation. Consider acrylic fiber production, where DMF dissolves the polymer cleanly and lets it be spun into threads. Chemistry at work: the specific arrangement of those atoms in DMF delivers that high solvency power. Pharmaceutical labs tap its properties to assist in creating active ingredients. Electronics manufacturers rely on its stability for printing circuits. Taken for granted by many, the molecule supports multi-billion-dollar supply chains.
A simple miscalculation of C3H7NO means contamination or off-spec material. That matters. In my experience, each kilo shipped represents not just a chemical, but months of investment in sourcing methylamine, carbon monoxide, and precise process control. Tight control over every step assures our downstream users—often facing little margin for error—that their end products won’t fail due to a basic ingredient.
This formula, though concise, comes with safety challenges. Nitrogen and oxygen in the same backbone make for a high-boiling, water-miscible, polar aprotic solvent. Benefits are plenty, like dissolving a wide variety of compounds, but user exposure must be managed. I have seen shifts in regulation that mean process adjustments overnight—for instance, strengthening vapor recovery and tracking emissions at every valve. As a chemical manufacturer, we do not get a pass if our formula handling contributes to poor work environments or ecological risks. This means keeping every operator trained, maintaining up-to-date analytics, and investing in closed systems.
Our work with the DMF molecule makes it impossible to ignore how global supply and safety depend on transparency. Every time we declare C3H7NO content, labs go to work validating that claim. Customers ask about purity, by-products, origins of feedstocks. There’s no shortcut; those five letters mean trust. Mislabeling or hiding process shortcuts might save money for a month, but brings regulatory and reputational disasters that take years to recover from.
Continuous improvement forms the core of our operations. Better analytics, stronger purification, and feedback from partners all play into making sure every shipment of DMF upholds the reliability embedded in that simple formula. Regulations grow stricter, and the push for greener chemistry grows louder. We look at new catalyst systems, cleaner energy approaches, and more robust solvent recovery, striving to make this molecule as responsibly as possible.
In the end, the formula C3H7NO anchors decades of research, production, and innovation. It guides reactor design and influences how we train teams and update safety systems. Even though the formula looks simple on paper, its performance keeps entire industries moving. For us, each order filled is a promise kept: careful production, reliable supply, and constant adaptation to a changing regulatory and technical landscape.
As a manufacturer who handles dimethylformamide daily, I've seen how small decisions around storage can mean the difference between smooth production and serious operational delays. If you've ever noticed the sharp odor of DMF escaping from a drum or witnessed oxidation tainting a batch before reach the reactor, you know how much proper storage matters.
DMF reacts to careless handling by picking up moisture and, over time, breaking down into formic acid and dimethylamine. Moisture sneaks in fast, especially in warm, humid settings. This might not sound dramatic, but it can corrode steel drums or change the pH enough to spoil sensitive reactions. Chemical purity drives our process yields—so keeping DMF dry, air-tight, and cool pays off on more than just lab reports.
We store DMF in dedicated, clearly labeled containers made of steel with proper linings or high-density polyethylene. The right choice hinges on how fast you'll use up inventory. Most of our drums don’t last more than a few months before hitting the plant floor, but even in that short window, condensation and air exposure can creep in if seals get damaged.
I instruct my team to check each drum on arrival for dents, corrosion, or loose bungs. Damaged drums let in air or water vapor. If this happens, the smell gives it away, but by then contamination has started. Moving damaged stock to a separate zone prevents cross-contamination down the line.
Temperature swings push vapors around and increase the risk of pressure buildup. Our solution is simple—DMF sits in a cool, well-ventilated warehouse, away from sunlight and sources of ignition. We calibrate storage temperatures by keeping drums out of direct heat and maintaining a dry environment using silica gel or sealed rooms with dehumidifiers during the rainy season.
Good air quality isn’t just about safety compliance; it helps prevent odors from escaping and protects the health of our team. Fume hoods and local exhaust near transfer stations keep DMF off the production floor so headaches, sore throats, and worse don’t start turning up in the shift logs.
Storing DMF means accounting for pressure changes. Fitted vent caps avert dangerous pressure buildup but must stay intact to avoid leaks. We log every opening and transfer. The few minutes saved by skipping vent checks can cost hours in cleanup if a drum leaks or vents unsafely.
There’s temptation to top up drums with leftover material, but this practice leads to inconsistent quality. Freshness counts. We assign lot numbers and “first-in, first-out” rotation to keep materials moving. Once opened, a drum moves to use within days instead of weeks, reducing exposure risk.
As newer employees join and processes evolve, I stress hands-on training paired with updated signage and practical drills. Guidelines change as we adopt new storage options, automate temperature checks, and improve containment in case of a spill. Audits and real incident reviews shift our methods from textbook to practical solutions.
From the manufacturer’s side, storage isn’t just box-ticking. Smart handling, constant oversight, and old-fashioned attention to detail keep DMF safe, pure, and ready for production—long before any paperwork starts moving.
Sitting at the helm of production, you witness how a solvent like dimethylformamide, or DMF, touches every part of the workflow. From blending to storage, DMF demands respect, not only for what it unlocks in synthesis but for the risks it brings. We’ve worked through small spills, rushed turnarounds, and the relentless pull of meeting deadlines. Each time, the story ends the same way: those who ignore the basics get bitten. The stakes spike for the floor crew and the tech team alike. A single shortcut—leaving a valve cracked, skipping the gloves—can cost days in clean-up and put health in real danger.
Experience teaches fast: DMF passes through skin with surprising ease. We’ve learned that not every glove resists it for long. Simple latex breaks down under constant use. In our runs, only nitrile or neoprene hold up for shifts. Even with top-quality gloves, changing them regularly keeps the nagging worry of absorption at bay. Eyes come next. Splashes don’t announce themselves. Goggles make a difference. One senior operator spent a week on medical observation after a stray drop made contact. Since then, face shields sit alongside every batch line. Despite daily repetition, vigilance never feels old fashioned.
The odor of DMF leaks out easily. At scale, ventilation isn’t window dressing: it cuts fumes at the source. Years ago, a ventilation shutdown forced evacuation. That event pushed us to double up on airflow, with ducts close to any open vessel and frequent filter checks. Open windows and fans don’t cut it; only robust extraction stands between comfort and headaches. Respirators? They aren’t just for emergencies. Any direct work over a tank calls for fit-tested masks—not the simple dust covers, but organic vapor cartridges made for the long haul.
Even off shift, DMF keeps working. Drums must stay away from sparks, away from acids and oxidizers. Wet floors have ruined more than one pallet: water reacts, so dry storage matters as much as temperature. We track signs of swelling or corrosion on drums with every inspection. Early minor leaks save days of disaster later. Even sealed drums give off trace vapor. That pushes clear labeling, secondary containment, and quick removal of any mystery container. Every team member knows if a drum turns up without clear marking, it doesn’t move until checked well.
The right paperwork on a wall means little without hands-on drills. We send new hires straight into mock scenarios: spills, glove tears, eye-wash runs. They don’t just read instructions—they practice with the real kit until reflex kicks in. Veterans keep their edge with refreshers. Management catches issues early by walking the floor, not from an office chair. Every near-miss sparks group review; the point isn’t blame, it’s teaching the next person in line what to do better. This kind of investment turns basic caution into embedded culture.
No shortcut replaces the hard-won knowledge earned on a real production line. We treat dimethylformamide with the respect any powerful tool deserves. Keeping people safe takes patience, training, and honest reevaluation of habits that might feel comfortable but break down under stress. Real safety grows from choice and repetition, not from warnings on a label or a distant directive. In the end, that mindset defines whether a manufacturer truly stands behind its people and its product.