Factories first churned out dinitrotoluene over 150 years ago, prompted by the world’s shift from black powder to safer, more powerful explosives. As a chemical manufacturer, I’ve walked the shop floor where generations before me set up crowded nitrate towers, the air humming with the scent of nitric acid. The earliest days focused on production for munitions—our grandfathers needed dinitrotoluene to feed the hunger of war. Over time, demand grew from paint and dye makers who saw the value in its chemical backbone. Stronger standards for purity soon followed, transforming what once was a crude arms ingredient into something critical for foams, elastomers, and coatings. Today we’re not just inheritors of a recipe; we’re stewards of hard-earned lessons in process safety, chemical handling, and risk management. Every pump and pipeline reflects history’s trial and error.
Talking about dinitrotoluene in a manufacturing context always starts with its role as an essential intermediate. In bulk chemical plants like ours, we handle mixed isomers, with 2,4- and 2,6-dinitrotoluene forming the core. These aren’t obscure catalog items—they underpin material classes found in everything from seat cushions to automotive coatings. Our lines deliver thousands of tons annually, each batch designed to tight technical standards, enforcing reliability for downstream polyurethane makers. What sets industry-scale dinitrotoluene apart from lab-grade or small-batch equivalents is the relentless drive for purity and consistency in every feedstock drum that leaves our facilities.
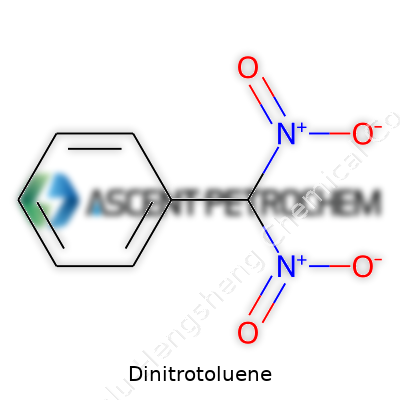
At room temperature, dinitrotoluene settles as yellow crystals, holding a faint odor reminiscent of sweet phenol. Melting points cluster between 68°C and 70°C for the favored 2,4-isomer. Water doesn’t dissolve it worth mentioning, but it loves organic solvents like acetone and benzene—a trait we use to plan cleaning cycles and recovery steps in our units. These same properties complicate handling; dust can collect in slow corners, so we keep scenes ventilated and minimize spill potential. Its chemical backbone combines nitro groups on a toluene ring, conferring both reactivity and stability. High enough temperatures or the wrong catalyst can push it toward decomposition, so we design reactors for fine control. In real-world operations, vapor and dust control shape everything from PPE choice to the gauge on a burst disk.
Each tank shipment or drum reflects more than just a CAS number and a purity figure. As a long-term producer, we document isomer ratios, moisture content, acid numbers, residual mononitrotoluene, and color values alongside required hazard labeling. Over the years, user feedback from the foam industry has pushed us to find better ways to drive down trace contaminants. One incident where over-acidic product fouled a customer’s prepolymer batch led us to install inline neutralization and real-time monitoring—no spreadsheet or spec sheet replaces the feedback loop from practical users. GHS and REACH rules require visible hazard warnings and clear transport codes, and every packaging line includes quality checks to prevent cross-contamination or mislabeling. The regulatory landscape grows stricter each year, and our document control office expands to keep pace, guided by lived compliance experience, not just templates.
We synthesize dinitrotoluene through stepwise nitration of toluene—first to mononitrotoluene, then further nitration to dinito derivatives. This classic process seems simple on paper but brings a pile of safety concerns in reality. The exothermic reaction, generation of acid mist, and accumulation of byproducts require engineered vessels, precise controls, and disciplined training. Our reactors use jacketed cooling, automated acid dosing, and regular sampling to catch anomalies. Operators credit years of practice for unblocking lines before they solidify and responding to alarms conditioned by real incidents, not by theory. Any deviation risks runaway scenarios, so safety interlocks and rigorous standard operating procedures anchor the process. Downstream purification relies on multi-stage washing, neutralization, and solvent extraction, each tailored over time to the needs of our main customers. Years of collaboration with process engineers and end-users led to present routines for waste acid reuse and effluent neutralization, helping lower costs and emissions.
In the hands of an experienced plant team, dinitrotoluene becomes a versatile substrate. Downstream, hydrogenation converts it to toluenediamine, a key polyurethane precursor. Here, trace metal residues or isomer impurities can produce costly off-spec runs, so we invest in closed-system transfer and tight process analytics. We’ve seen technical service chemists tweak conditions to test novel polyurethane grades or new polymerization catalysts. Some external partners oxidize it to related acids or reduce it selectively for complex syntheses. Decades of process optimization taught us that even minor impurities can poison a catalyst bed or bias an aromatic substitution, so feedstock control never rests. In research collaborations, modified dinitrotoluenes serve as tailored monomers, but large-scale production remains anchored to classic hydrogenation and nitration chains. Each operational shift teaches something new—how subtle changes in reaction temperature or agitation shift yields over days or months.
Dinitrotoluene goes by several trade names and shorthand forms—DNT, 2,4-DNT, and 2,6-DNT, most often. Some legacy users still request “dinitro derivatives” or “yellow oil” by old plant codes, but clarity matters more now. Within regulatory documents, our labeling teams favor IUPAC names, but most technical staff and customers default to DNT and specific isomer numbers, which keeps orders unambiguous. Confusion between isomer types has caused production hiccups for careless buyers—a mix of “2,4” and “2,6” won’t suit most processes. For export, customs requirements force another layer of nomenclature and hazard classification, so every shipping manager juggles half a dozen codes and standards before a batch departs.
Dinitrotoluene’s hazards can’t be shrugged off. Chronic exposure links to health risks, so our teams live by strict PPE routines: double gloves, chemical splash goggles, and dust-resistant suits. Engineering controls keep concentrations below occupational limits, and every spill triggers formal response protocols. I recall the old days when patching a small pump leak passed as “good enough”—the industry learned better after costly incidents and regulatory crackdowns. Modern standards lean on comprehensive ventilation, real-time air monitoring, and formal medical surveillance, all rooted in past experience and incident data. Mitigating environmental discharge involves acid scrubbing, water treatment, and tested emergency containment. Site-wide safety meetings drill everyone on evacuation, first aid, and the risks of static electricity. Training now means real field exercises and deep familiarity with toxicology, not just slide decks. It’s one of those chemicals that leaves you constantly vigilant and negotiating between productivity and safe operation.
Though once a staple for munitions, today’s dinitrotoluene finds far broader use in materials science. Over half our annual output funnels directly into polyurethanes—soft foams in automotive seats, rigid insulation panels, elastomeric wheels, and even adhesives. Paint and pigment producers rely on its structure for designer dyes. Specialty explosives use persists, mostly in small-volume civil engineering and demolition. Decades of market evolution taught us to support formulation trials and scale-up processes alongside clients, offering feedback on how distillate purity or specific isomer blends perform. Our experts regularly field questions on compatibility with new catalysts or downstream reagent blends, and we invest in pilot trials for evolving applications. Extended market relationships grew from a disciplined effort to adapt grade offerings to changes in foam chemistry or textile standards.
Innovation thrives at the intersection of practical knowledge and academic insights. Over the years, our plant invested in joint research with polymer groups, testing new dinitrotoluene-based monomers for resilience or flame retardance. Academic partners bring fresh analytical techniques or greener synthesis routes, while our staff offer the hard-won expertise of scaling up from flask to railcar. Recent research trends push us to reduce byproducts, limit process waste, and find catalysts that boost selectivity. Ongoing projects examine continuous-flow nitration and improved solvent recycling. As regulatory and public pressure for sustainability increases, we’ve retooled aging plants with waste heat recovery and closed-loop washing cycles, guided by data and collective experience, not just environmental rhetoric. R&D at the factory level hinges on daily dialogue between lab chemists, operations staff, and end-users—a network that grounds progress in real-world needs.
Dinitrotoluene brings clear health and environmental hazards, backed both by regulatory science and our own experience. Chronic exposure links to nervous system effects and liver stress; acute exposure risks headaches and skin irritation. I’ve seen the difference that personal diligence makes—plants with strong hygiene and PPE records see fewer long-term issues. All new employees receive thorough toxicology briefings. Routine air monitoring, urine tests, and medical checkups became standard not out of compliance box-ticking but after watching former workers struggle with undiagnosed illnesses. Environmental managers supervise water and air emissions, and accidental spills trigger mandatory reporting and remediation. Upgraded filtration and effluent controls cut our discharge levels sharply in the past decade, showing that diligence and investment measurably improve plant safety.
Every year brings new questions about the future for dinitrotoluene in manufacturing. Customers push for greener sourcing, cleaner process water, and lower emissions. We face rising scrutiny over production waste, occupational risk, and long-term health outcomes. Some see opportunity in bio-based feedstocks or new catalytic routes; I see years of investment and training, matched by technical hurdles that defy easy fixes. Digitization and real-time process controls promise tighter specs and safer operation, but only if paired with hands-on experience and clear accountability. Expanding applications—advanced foams in green construction, specialized polymers for electronics, lower-toxicity derivatives—drive steady R&D commitment.
Our teams remain cautious optimists. The basic backbone of dinitrotoluene chemistry fits the world’s need for versatile, cost-effective building blocks, but adaptation defines survival. Future markets will likely reward those who combine process rigor with investment in greener solutions, guided by memories of what old methods got wrong and new needs demand. In this, the chemical manufacturer’s wisdom matters as much as any new patent or imported process. Our story with dinitrotoluene isn’t just making a commodity—it’s about shaping its safe, sensible future.
Walking past reactors loaded for Dinitrotoluene processing, most visitors only notice the yellow-tinged crystals and the stringent safety signs. In our line of work, that yellow powder isn’t just another chemical; it stands as one of the main building blocks for both industrial progress and, occasionally, controversy. Having spent years producing Dinitrotoluene, I’ve watched the ebb and flow of demand connected to changes in global economics, safety standards, and technological advancements. Its core value stems from versatility coupled with what it enables downstream in production.
Polyurethane foam shows up everywhere: insulation panels inside homes and refrigerators, seats and dashboards in vehicles, mattresses, and shoe soles bouncing underfoot. This common thread starts with Dinitrotoluene. Manufacturers rely on nitroaromatic chemistry to kick off the reaction creating toluene diisocyanate. Without Dinitrotoluene, domestic and automotive insulation would look very different. Spare parts that need durability and resilience benefit from rigid and flexible polyurethanes. Production facilities have optimized Dinitrotoluene conversion rates to meet swelling demand, particularly in booming construction and automotive sectors. Quality control teams test each batch thoroughly because poor feedstock can halt an entire line, raising costs and wasting energy.
Every operator working with Dinitrotoluene understands the associated risks. The compound requires care to prevent inhalation and skin contact. We invest in updated protection equipment, real-time air quality monitoring, and rigorous training for every new worker. Experience has taught us that even a lapse in procedure can have real-world health consequences. Strict documentation and process auditing help us stay in compliance with evolving European and international limits. Regular medical checkups for staff help us spot problems early, so we can safeguard employee well-being before regulatory penalties ever come into play.
Dinitrotoluene doesn’t just make it into foam. During past decades, its energetic properties attracted attention from the explosives sector. Used as a component in industrial blasting gels, Dinitrotoluene blends well with other explosives. Few people realize this application persists for certain mining or construction needs, but environmental and safety pressures continue to shrink its share in that arena. Instead, we see chemical production taking the front seat, as market forces push for more sustainable production methods and lower-impact derivatives.
Sourcing raw materials for Dinitrotoluene means working closely with upstream suppliers to minimize impurities and reduce volatile organic emissions. Our team adopted process upgrades such as closed-loop systems that capture exhaust, reducing both odor and community complaints. These changes triggered capital projects and required management buy-in, but they pay off. Neighbors appreciate reduced emissions, and safety bodies conduct audits with greater confidence in our systems. Focused research into green chemistry methods is driving incremental progress, even though scaling up these solutions can take years.
From every shift, it’s clear that demand for Dinitrotoluene rides on the back of changing markets and tougher safety standards. Raw material volatility and regulatory tightening will keep producers on their toes. Listening to frontline staff, investing in reliable equipment, collaborating across departments, and never taking shortcuts has shaped this plant’s reputation. Industry can’t run without raw materials like Dinitrotoluene, but the way forward demands constant self-reflection and readiness to refine both processes and priorities.
In the business of synthesizing Dinitrotoluene (DNT), the subject of safety never slips off our radar. Plenty of stories circulate about chemicals and their dangers, but DNT deserves attention based on true industry experience and verified science. Nobody in our field can ignore the potential health hazards of DNT—ignoring them carries real-world consequences for our workers, the communities nearby, and the end-users of downstream products.
Dinitrotoluene’s primary value rests in its role as a precursor for toluene diisocyanate, a key building block for polyurethane foams and elastomers. Handling ton-scale volumes in enclosed reactors brings routine exposure risks, and decades under our belts stress the importance of strict engineering controls and compliance with occupational limits.
We see acute health effects if someone accidentally inhales DNT vapors or absorbs it through skin. Workers report headaches, dizziness, and proper medical monitoring points to risks for anemia from repeated exposures. The skin readily absorbs DNT, which makes even minor spills a workplace priority. The yellow discoloration and skin irritation are warning signs everyone in the plant recognizes instantly.
The International Agency for Research on Cancer gives DNT a Group 2B classification—possibly carcinogenic in humans based on sufficient animal data and limited human evidence. We don’t dismiss the fact that long-term or repeated exposure shows a link with increased risk of some cancers. Researchers studying workers with years of exposure have found higher-than-normal rates of bladder cancer, giving weight to the association.
In our facilities, effective risk management starts with process design and reliable containment. Closed systems, scrubbing units, and rigorous maintenance keep everyday exposures far below legal limits. Regular industrial hygiene programs run air sampling campaigns and medical checks to spot trends before they become problems. When process upsets or leaks happen, personal protective equipment is never optional—respiratory masks, gloves, and chemical-resistant suits are standard kit on every line handling DNT.
Transport and storage also require constant vigilance. Our teams use dedicated tankers, atmospheric monitoring, and real-time data to prevent accidental releases. Spill kits, emergency drills, and water management systems stay close at hand because a single incident could impact not just the staff, but also downstream water bodies if DNT enters wastewater streams. Worry over environmental persistence motivates us to invest heavily in advanced wastewater treatment so DNT and its breakdown products don’t linger in the environment.
The public often sees headlines about chemical accidents and asks whether DNT is “worth the risk.” The answer lies in how companies operate, not in simple yes-or-no questions. Responsible manufacturers encourage honest reporting, invest in training, and hold plant leadership accountable for safety culture. We listen carefully to regulatory agencies and regularly share best practices through industry associations—peer learning has saved lives more than once.
Every year brings new research and stricter guidelines. We consistently reevaluate exposure limits, invest in process upgrades, and look out for safer alternatives. DNT isn’t a toy, and we treat it with deep respect. For those on the ground floor, health and safety are part of the fabric of daily work, not just items on a checklist. Experience, science, and genuine responsibility shape how we respond to the risks—not scare tactics or wishful thinking.
Producing and handling dinitrotoluene brings a specific set of responsibilities. Anyone involved in actual synthesis and storage of this chemical knows real-world constraints—these aren’t best-case scenarios written in clean offices, but measures honed by keeping workers, facilities, and communities safe for decades.
Dinitrotoluene, used extensively in polyurethane manufacturing and explosives, requires extra attention in storage. Its chemical characteristics demand respect. Unlike some raw materials, this compound reacts to heat, moisture, and light with unpredictable vigor. The yellow, oily liquid seems benign on inspection, but it can create real hazards if untreated or mishandled. On busy sites, teams battle with factors like aging roofs, fluctuating temperatures, and the human tendency to cut corners for convenience. Years in manufacturing dinitrotoluene teach hard lessons—heat can sneak up fast, condensation forms in the unlikeliest corners, and small spills tend to grow if left as tomorrow’s problem.
Controlling temperature sits at the top of any list, but the reality is more complex than setting a thermostat. The compound degrades with heat, so production spaces use insulated tanks, shaded warehouses, and forced ventilation. Automated temperature monitoring systems can make all the difference between stable inventory and a major incident. Leaks or poorly insulated valves might drive up local temperatures within specific pockets, becoming hotbeds for decomposition or fire risk.
Moisture brings its own dangers. Dinitrotoluene and water do not mix well; persistent condensation on containers, unexpected rain blowing through open doors, or damp pallets all increase risk of contamination and dangerous reactions. Most facilities rely on sealed steel drums or lined bulk tanks with pressure-relief features, keeping water at bay and helping manage any slight increases in internal pressure during storage.
Vapors from this chemical do no one any favors. Good ventilation sounds simple, but the truth is, industrial fans only help if teams keep all doors and louvers clean and unblocked. Over time, we’ve learned grouping dinitrotoluene containers away from oxidizing agents, food, and common workspaces pays off. Chemical segregation prevents accidental combinations and underscores focus on worker safety. Many facilities employ separate fire-rated storage rooms, a practice born from bitter experience.
Complexity breeds error. Over years, most manufacturers see the benefits of written protocols, clear labeling, and regular training. Nobody can rely on memory alone in a storeroom with dozens of similar containers. Repeated drills for spill containment and evacuation aren’t box-ticking exercises—they cut down reaction time if something goes wrong. We see the payoff in low incident rates, steady product quality, and fewer insurance headaches.
Continued investment in storage infrastructure turns out cheaper than accident cleanup or regulatory fines. Pressure to cut corners surfaces in every cost discussion, but experienced manufacturers rarely gamble on this front. Collaborating closely with environmental health experts, scheduling frequent equipment checks, and applying lessons from industry mishaps create long-term value far beyond simple compliance.
The work isn’t glamorous, but it keeps doors open and people safe. Dinitrotoluene’s risks never disappear—the challenge is consistent discipline. In this business, lighting, ventilation, training, and attention to detail matter on a day-to-day basis, not just on paper.
Any chemical manufacturer working with dinitrotoluene (DNT) develops an appreciation for the logistics that keep this material moving safely from plant to customer. DNT, with its utility in producing polyurethane foams and other industrial products, requires robust transport measures. Getting safety protocols right is essential for our employees, carriers, and the communities along the route. The work starts with a clear understanding: dinitrotoluene has moderate toxicity and presents a serious fire risk, so every stage of storage and transit must run according to rigorous protocols.
Bulk shipments of DNT move in tank trucks or railcars designed to resist corrosion and prevent leaks. Our experience tells us ordinary tanks don’t cut it. Stainless steel or specialized coatings prevent chemical reactions that could compromise the tank or contaminate the product. Fittings, seals, and relief valves—routine parts for most chemicals—take on extra meaning in a DNT operation. Every part undergoes regular inspection and replacement. Any shortcut increases the chance of costly spills or, worse, a dangerous incident.
The most reliable systems start with people. Our warehouse and logistics crew need regular training on emergency response, spill containment, and correct loading practices. We hold drills, quiz our staff on the latest updates, and never settle for surface-level compliance. Safety checklists are only useful when people understand why each step matters. DNT fumes can cause health problems, so loading and unloading always happen in well-ventilated zones. Our process engineers keep an eye out for small leaks or curious odors—nothing escapes notice.
The paperwork might look tedious, but it keeps suppliers, haulers, and end users on the same page. Each shipment carries a manifest specifying the DNT quantity, concentration, hazards, handling instructions, and emergency procedures. Drivers receive dedicated training in hazardous materials law and undergo regular audits. Routes avoid densely populated or environmentally sensitive areas as much as possible, and GPS tracking lets our operations center monitor every shipment’s progress and respond to any hold-ups.
Our own standards draw on industry best practices and lessons learned from the rare accidents that have happened elsewhere. Past chemical incidents often trace back to missed inspections or neglected maintenance. Investing in sensors that monitor pressure, temperature, and vapor release inside tanks gives early warning of trouble. Each time a shipment finishes without incident, that outcome means the system worked as intended—a balance of safe design, clear communication, and professional pride.
Transporting DNT safely never counts as “done.” New research, technology advances, and regulations push us to review our methods and keep improving. We have open lines with regulators and emergency services to share updates and gather the newest guidance. Safe delivery relies on a culture that encourages people to speak up if something seems off, no matter how small. In a practical sense, that culture translates to lower risk for everyone involved and helps keep production flowing smoothly.
We produce dinitrotoluene (DNT) every day in our facility, and we know its chemical formula by heart: C7H6N2O4. These numbers and letters represent more than a simple code—they echo through every stage of our production process, define how we handle raw inputs, and shape the downstream uses of the product. Dinitrotoluene has six hydrogen atoms, seven carbon atoms, two nitrogen atoms, and four oxygen atoms. Each of these elements demands respect when combined. It’s vital to grasp the implications of that formula when considering the material’s behavior. From storage and handling to safety protocols, this chemical’s formula is at the center of our practical decisions.
Every operator, chemist, and supervisor on our line learns to appreciate the significance behind C7H6N2O4. Dinitrotoluene doesn’t forgive mistakes in handling or measurement. Nitrogen and oxygen in the molecule tell you immediately that this is not something to take lightly; potential reactivity, thermal decomposition points, and inhalation hazards trace straight back to those atoms. Knowledge of the formula influences everything from ventilation setups in our blending rooms to the thickness of gloves. Many articles talk about chemical formulas as if they are trivial details, but in manufacturing, ignorance leads to problems—expensive, dangerous ones.
We anchor our safety training and risk assessments in that formula. Dinitrotoluene boils at a moderate temperature, and its nitro groups increase sensitivity to heat and contaminants. Spills are prevented by containment systems built for materials containing nitrogen oxides. As a manufacturer, we devote significant resources to regular equipment checks and staff education. Because two nitro groups and a methyl ring spell out risks: potential for toxic vapors, implications for chronic health exposure, and heightened reactivity during storage or processing. These aren’t theoretical risks, and experience has taught us the value of vigilance.
We see different isomers of dinitrotoluene, such as 2,4-DNT and 2,6-DNT, differentiated by the placement of nitro groups on the toluene ring. Small changes in atomic arrangement impact melting points and solubility, which affect both safe processing and the end use. For the explosives sector, for example, precision at the molecular level has practical consequences; impurities or errors in synthesis can lead to unstable products. In our plastics intermediates operations, consistent molecular composition keeps batch quality high, so knowledge of the formula runs through our QC reports.
Maintaining a reliable supply of dinitrotoluene means always being aware of the regulatory shifts targeting nitrogen oxide compounds and monitoring emissions down to a fine degree. The chemical formula serves as a reference point for compliance teams overseeing inventory tracking or environmental reporting. As environmental scrutiny tightens worldwide, we continuously modify our processes to reduce exposure and waste. Solutions focus on closed handling, advanced filtration, and secondary containment—rooted in a full understanding of C7H6N2O4’s potential pathways in the environment and workplace.
From our plant floor to the boardroom, respect for dinitrotoluene’s chemical makeup drives practical decisions. It’s not just about memorizing a chain of letters and numbers. It’s about connecting chemical structure with real-world risks, production realities, and ways to build a safer, more efficient operation. Science lives in these details, helping us make better decisions every shift, every shipment, every audit.