Methyl ethyl carbonate (MEC) has drawn interest from chemical producers over recent decades, tracing its roots to the branching path of alkyl carbonate research in the 20th century. Early-stage research focused on simpler dialkyl carbonates—methyl and ethyl forms—due to their use as solvents and intermediates, but increasing demand for custom-tailored molecular structures pushed innovation towards mixed carbonates. Our decision to scale up MEC came in response to growing requirements from battery developers and specialty solvent formulators who recognized MEC’s unique balance of polar and non-polar character. This shift from lab curiosities to industrial mainstays reflects the practical needs of downstream industries seeking alternatives that outperform classic carbonates and mitigate regulatory pressure on legacy solvents.
At our plant, MEC emerges as a clear, colorless liquid, with a mild, slightly sweet odor. We constantly monitor purity because even traces of water or byproducts spoil product consistency, which in turn affects downstream application. The demand comes largely from high-performance electrolyte blending—a sector with near-zero tolerance for contaminants. Our customers measure success in parts per million, so our process focuses on repeatable results, not just average values. Every drum, tank, and sample tells a story about process control, logistics, and how closely the workforce stewards quality from batch to batch. Seeing barrels labeled MEC rolling down the loading dock, we recognize the responsibility that comes with even a liter’s worth of material headed towards demanding markets.
MEC’s combination—molecular formula C4H8O3—offers a delicate balance. The boiling point stands just above 100°C, which sets it apart from both dimethyl and diethyl carbonates, making it more practical for applications where volatility must be tightly managed. The solvent power extends towards a range of non-aqueous systems, a quality that found appreciation with battery makers who chase high dielectric constants for electrolyte formulations. The compound dissolves in most organic solvents, but we track hygroscopicity with vigilant care, knowing that even atmospheric humidity changes physical properties in subtle ways. Flammability and vapor pressure enter the daily safety briefings. Plant engineers constantly manage those realities, applying mitigation strategies field-tested by decades of facility operations.
Labeling standards matter more than most outsiders recognize. Supply chain compliance not only means listing standardized names and hazard pictograms but also managing batch traceability for quality audits. Our team fields regular queries on purity, and the certificates we sign reflect months of work in calibration, with specification sheets distilled from customer dialogue. Typical results land well within the top percentile for known impurities: water, methanol, ethanol, and acetone. Any anomaly leads to overtime in our labs until root causes get identified and contained. Packaging choices stretch from drums to bulk tankers, each cycle integrating ever more robust tracking technologies reflecting international transport standards.
Commercial production of MEC favors reaction of ethyl alcohol and methyl alcohol with phosgene or dimethyl/dietyl carbonate transesterification routes. Phosgene-based synthesis lost traction due to worker safety, so our shift to phosgene-free methods reflects both environmental leadership and practical risk management. Process control means relentless attention to catalyst recycling, temperature modulation, and residence time. Continuous monitoring of reaction effluent helps in predicting heat spike potential or catalyst poisoning. We spend resources on residue recovery, solvent recycling, and effluent minimization to ensure cost-effectiveness and regulatory compliance. Engineers working on the lines know that process stability means profit, waste, and safety all hinge on a thousand little details embedded in a day’s work.
In the chemical synthesis community, MEC earns recognition for its flexibility. It provides a starting point for carbonylation, transesterification, and selective hydrolysis reactions, supporting the synthesis of more complex organic molecules. We frequently supply research labs exploring new cathode material electrolytes or pharmaceutical intermediates. MEC’s reactivity profile lets it function as an intermediate for carbonate exchange, underpinning newer, safer battery chemistries and plasticizer developments. In every shipment, downstream chemists count on our ability to maintain minimal impurity drag, because these carry over into subsequent reaction sequences. We track feedback from customers who push MEC through challenging organic syntheses, logging these lessons into our process refinement plans.
The landscape of nomenclature features more than one path to MEC. Global trade references terms like ethyl methyl carbonate, EMK, or CEMC. Some documents cite IUPAC designations, others stick to common trade names, depending on regional convention or end-use application. In the factory and during export clearances, accurate terminology prevents costly delays. Knowing what a client means by EMK or similar variations helps our logistics teams avoid errors that cascade through loading docks and customs paperwork.
Manufacturing MEC puts hazard management at the top of every safety protocol. Despite appearing unassuming, MEC carries risks—flammability, inhalation, and skin contact hazards present real challenges during bulk handling. Our protocols demand appropriate PPE, real-time gas analyzers around pumping stations, and fire suppression upgrades in storage areas. Annual audits by internal and external safety teams stress emergency response readiness and system redundancy. Employees undergo customized training because real experience on plant floors teaches nuances that textbooks skip. Knowledge of spill management, confined space entry, and process upset response comes from field drills, not paperwork alone. Compliance with GHS and REACH regulations forms a baseline, audited by our dedicated QA and HSE professionals who operate with lived responsibility for every gram manufactured.
Across the sectors we supply, energy storage innovation drives MEC demand. Lithium-ion battery manufacturers treat it as a crucial co-solvent in non-aqueous electrolytes, where the compound’s dielectric properties and stability at elevated voltages help facilitate rapid ion transfer and improved battery cycle life. Outside energy storage, laboratories employ MEC in specialty synthesis, chromatographic analysis, and as a reaction medium for fine chemical and pharmaceutical intermediates. Other uses include coatings development—where its solvent properties aid in viscosity adjustment and fast film formation. Our knowledge base grows with each new customer challenge; we routinely consult on custom blends and troubleshoot field performance, translating laboratory insights into practical plant solutions.
Over the years, our R&D unit invested in refining MEC production. Each improvement began as a challenge from a client or a sharp-eyed operator noticing variability in output. Working alongside academic collaborators, we have examined ways to increase catalyst life cycle, shrink unwanted byproducts, and recover residual alcohols more efficiently. Our data logged across multiple years shows steady gains in both yield and consistency. Insights gained from field deployment in battery labs feed into pilot runs, revealing process tweaks that can be scaled with confidence. Partnerships with clients in new market segments constantly shape our development priorities, giving our scientists a direct line of sight into practical problems and aspirational goals.
Worker and downstream user safety stand at the core of operational planning. Our toxicology reviews draw not only on regulatory canvassing but also on workplace health monitoring and independent academic studies. MEC exhibits low acute oral toxicity in mammalian tests, but prolonged inhalation or direct skin exposure raises concerns, especially in high-volume production lines. Over the years, routine health screening and real-time exposure monitoring have shaped our facility design and protocol architecture. Collaboration with industry partners and research groups supports ongoing evaluation of chronic exposure effects. Publicly available studies and confidential internal data supplement our safety data sheets and incident response plans.
Every forward-looking analysis on the plant floor reflects the unstoppable tempo of battery advancement, growing green chemistry mandates, and the unpredictable cadence of regulatory change. Battery innovation, electric vehicle proliferation, and new energy policies are expanding MEC’s potential market. We watch these trends closely, investing in both capacity expansion and process intensification. Every shift brings a new challenge—whether it is customers searching for ever-purer grades, researchers asking for tailored variants, or regulators ratcheting up safety and environmental thresholds. Our commitment looks beyond retaining current markets to exploring greener feedstocks and closed-loop process designs. In the evolving world of fine chemicals, the ability to adapt emerges directly from front-line experience shaping what comes next for MEC and all materials in its class.
A lot of folks in our industry are curious about methyl ethyl carbonate (MEC) and its role beyond chemistry textbooks. In our plant, we think about it daily—handling it, shipping it, and talking with engineers who need the real story about why MEC keeps showing up on their raw materials list. From a manufacturer’s angle, it’s easy to see which uses have staying power and which are still emerging.
Batteries have changed how manufacturers like us approach solvents. MEC has become a staple in electrolytes for lithium-ion batteries. We noticed that MEC stands out for its ability to enhance the transport of lithium ions between electrodes. It brings a low viscosity and relatively high dielectric constant, which contribute to faster ion movement. In practice, this translates to improved battery efficiency, better cold-weather performance, and longer cycle life. Most of the demand we see today comes from electronics and electric vehicle markets, both hungry for reliable, high-density storage. The cost and purity requirements are strict; we meet them by focusing on advanced distillation and in-line quality monitoring, not just lab testing after the fact.
Before batteries took over the headlines, MEC held a reliable spot in the solvent world. In specialty coatings and inks, its fast evaporation rate and solvent strength allow for thinner, more uniform layers. Paint chemists often call asking for batches with tighter boiling range and ultra-low moisture levels to suit performance coatings or certain printing processes. Some of our long-time customers make adhesives that rely on MEC’s balance between solvency and drying time, which helps avoid tacky finishes and supports high-speed production lines.
MEC gets picked as a chemical building block too. Several pharmaceutical and agrochemical producers order it for use as an intermediate, often in finely-tuned syntheses where small differences in raw material quality can derail an entire production batch. On our end, batch repeatability comes from strict process control and equipment maintenance rather than shortcuts. Reliable logistics matter just as much as product purity, especially for companies working with tight turnaround times.
Demands for higher purity are non-negotiable for battery manufacturers, but similar scrutiny applies for environmental and worker safety concerns. We follow guidelines that keep emissions in check and invest in closed transfer systems, so people and the environment stay protected through every stage of production. Real progress comes from steady upgrades rather than flashy one-time overhauls. Industry-wide talks about green chemistry have started showing up in large orders for high-purity MEC made with less waste and fewer energy inputs.
Supplying MEC has always required a careful balance between production costs and customer needs. Global sourcing of raw materials brings headaches: delays, price swings, and quality swings. We track our supply chain as closely as the reactors in the plant. We have learned to keep extra inventory on hand, tweak formulations based on real-time feedback, and invest in automation to minimize human error. As renewable energy grows, cleaner battery solvents matter more, so our R&D lab runs pilot-scale trials to cut down on process waste and boost yield. Customers expect transparent communication—they call us partners, not just suppliers, when we deliver on those promises.
People who work with chemicals every day see more than numbers and hazard symbols. Methyl ethyl carbonate (MEC) enters countless processes, especially in battery electrolytes and specialty coatings. Its vapor has a distinct odor, its transparency may fool those who rely on the senses alone, but trust experience—smooth operations demand full respect for the risks.
Methyl ethyl carbonate lands in the category of flammable solvents, so attention always starts at storage and handling. It evaporates quickly, and the vapors can catch fire if an ignition source appears. Labels warn of this, but real awareness comes after seeing a drum that vented on a humid afternoon or a flange leak traced with a solvent sniffer. Fire departments don’t take chances with solvents like these, and neither do those responsible for keeping a plant running.
Direct contact brings another challenge. Skin holds up well against many things, but liquid MEC can cause irritation after repeated splashes. Inhalation isn’t a friend, either—workers exposed to high concentrations report headaches or mild dizziness. Most exposures can be avoided with proper procedures, but shortcuts can lead to incidents that ripple out far beyond a single shift.
All chemicals come with safety documentation, but manufacturing traditions rely as much on stories as on written programs. Years spent on a busy line reveal a pattern: fatigue after a day spent venting and purging, rashes from poor glove selection, and the infamous panic that comes with vapor alarms. Regular reviews of air monitoring data picked up more in one chart than a year’s worth of incident investigations. Following the data, teams move to closed transfer and double-checked ventilation.
Methyl ethyl carbonate has a low toxicity compared to some other commercial solvents, but safe doesn’t mean harmless. People don’t bounce back from fire and chemical exposure just because guidelines say the material is “moderately hazardous.” Job rotations get reworked and protective gear gets selected by those who have seen the other side of minor lapses—lost time, stressed personnel, and expensive shutdowns.
Relying only on a printed hazard summary never covers all situations. On the shop floor, storing MEC upright and away from acids or oxidizers became policy after an exothermic mix-up caused a shutdown for days. Shift change briefings focus on long-term risks and lessons learned the hard way. Hazard reviews encourage staff to call out problems instead of keeping them quiet. Confidence to do this grows over years, not from a single day of training.
Plant teams use grounded drums, detailed ventilation checks, and flameproof lighting because those controls worked in the worst conditions. The best outcomes come from keeping exposure limits low—both by technical engineering and by empowering operators to stop a job the moment something feels off.
Methyl ethyl carbonate brings value in energy storage and specialty synthesis, but it keeps everyone honest about the reality of industrial risks. No solvent ever truly becomes routine. With every batch, teams shape their safety culture from open communication and repeated review. Lessons carved from real experience last longer than any memo taped to a wall.
People often ask us for straight answers about the chemicals we produce. Some want the CAS number. Some want the synthesis pathway or practical pointers for handling. Other times, it’s as direct as “what’s the formula?” Take methyl ethyl carbonate—one of those names that, despite sounding familiar, does not always get the immediate recognition outside the factory floor or laboratory. From decades of hands-on experience, I can say that answering questions like this shapes how the rest of the work unfolds, from R&D to shipping.
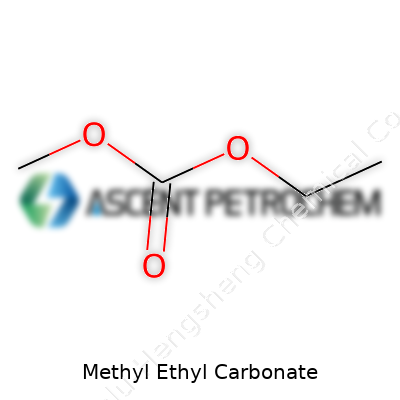
Methyl ethyl carbonate carries the formula C4H8O3. To break it down from a manufacturing angle: its structure stems from methyl (CH3), ethyl (C2H5), and a carbonate group. The molecule follows the general carbonate ester structure, with an oxygen linked between the carbonyl group and the two alkyl groups. This arrangement—one methyl and one ethyl group bonded to the carbonate—distinguishes it from more common dialkyl carbonates like dimethyl or diethyl carbonate. You see this formula come alive in the reactor, right where dimethyl carbonate and ethanol meet, in a transesterification reaction. From that point, every operational tweak, from temperature control to impurity management, depends on a clear understanding of this structure.
A precise chemical formula means a predictable molecule. That’s not just textbook knowledge; it’s vital to scaling up synthesis and controlling costs. Precision feeds into downstream applications. In battery electrolytes, for instance, knowing the reactivity and exact composition tells us about solvent properties—viscosity, dielectric constant, flash point. Small shifts in the ratio of methyl or ethyl groups can shift safety margins or change the performance profile in lithium-ion cells. Overlooking the basic formula can lead to unintended chemistry, contamination, or batch rejections. Quality doesn’t come from luck. In production, mistakes tie back to chemistry fundamentals more than anything else.
Getting the details right—starting with C4H8O3—sets everything in motion. From analytical chemists to engineers on the line, there’s no cutting corners. Analytical equipment like NMR or GC-MS works only as well as the standards and methods allow. If even one carbon atom slips in labeling, we risk invalidating hundreds of tests. It’s the difference between making material that partners trust and ending up with barrels nobody wants. This kind of reputation in manufacturing doesn’t just happen. It’s built through consistency, which first roots itself in chemical accuracy.
Questions about structure and composition often flag deeper process concerns. For us, publishing the right formula signals more than technical competence—it shows we hold ourselves to the standards industry expects. As commercial lithium-ion battery demand keeps rising, customers need to trust every drop of solvent they buy. Errors multiply quickly in production, so we double-check inputs, test outputs, and never skip the basics. Every lot that leaves the factory carries the same formula: C4H8O3. We built our reliability starting with the certainty that comes from the fundamentals—atom by atom, molecule by molecule.
Methyl ethyl carbonate brings undeniable value to manufacturing, battery electrolytes, and specialty chemistry. Its physical properties let us push the boundaries in new applications. But no matter how exciting the demand profile or how pure a lot of product runs, trouble begins if storage gets taken lightly. Too many overlook the risks involved with lesser-known carbonates until something goes off-course in the warehouse or drum yard. Most times, problems tie back to storage practices, and at the manufacturer's end, we see those outcomes firsthand—sometimes in spectacular fashion.
Methyl ethyl carbonate vaporizes quickly under normal conditions and can ignite if left near an open flame or strong heat source. This isn't theoretical; over the years, we've seen insurance adjusters walk through facilities wiped out by flash fires that erupted on hot afternoons when drums sat in the path of sunlight, nowhere near proper ventilation. The result? Lost inventory, regulatory headaches, and a mess for everyone involved. It only takes a single afternoon of poor judgment to cause years of fallout.
Our team safeguards the material with temperature control from day one. Storage works best between 15°C and 30°C, out of direct sun, and clear from the reach of forklifts that can rupture a drum. Atmospheric moisture near the chemical brings in another layer of problems. Years back, we noted slight cloudiness in inventory stored at the edge of a damp wall—water can contaminate an entire batch, leading to off-specification problems down the line. Moisture exposure also paves the way for corrosion in steel containers. That’s why our plant now limits carbonate exposure to humidity and uses tight seals on all containers.
Our experience confirms plastic drums fare best for methyl ethyl carbonate, provided they’re certified for contact with organics. Steel barrels, if coated, still carry risk if coatings fail or valves go unchecked over months of storage. We routinely inspect all closures and prohibit “top-off” practices where air gets sucked into a drum, even if just for topping up lost product. Time and again, this one shortcut invites air and moisture into the mix, making quality control a recurring liability.
Separation remains non-negotiable. Flammable solvents and reactive metals keep their distance. We never stack beyond two drums high—they topple, spills follow. Every incident on record comes down to a gap in alignment, labeling, or routine checks. Installing simple exhaust fans and regular drum rotations make vast differences in risk reduction. Our teams know no shortcut replaces vigilance.
Whether regulators or local fire marshals drop by, demonstrating responsible storage policies builds lasting trust. We keep documentation on climate control, inspections, and spill response processes open for inspection. As producers, delivering methyl ethyl carbonate is only half our job; keeping people, product, and property safe is the part we sign our name to every single day.
Methyl Ethyl Carbonate brings value as a solvent and specialty intermediate, but working with it means staying alert and following industry-proven precautions. On our manufacturing floor, it never pays to underestimate the hazards connected to organic carbonates, especially this one. Years of experience tell us that the line between safe routine and a serious incident can get crossed by a single shortcut.
The first thing that becomes habit in our facility: gloves and goggles go on every time. Methyl Ethyl Carbonate will irritate skin and eyes, even if a splash seems small. Stubborn gloves or a fogged visor tempt people to cut corners, but those few seconds in PPE mean the difference between an uninterrupted shift and a trip to the wash station. Chemical-resistant gloves, splash-proof goggles, and lab coats or coveralls—these are standard for everyone touching product containers. In high-volume transfers, we upgrade to face shields and full suits. PPE doesn’t guarantee invulnerability, but it stacks the odds in your favor.
People new to the compound often underestimate the impact of vapors. Without airflow, Methyl Ethyl Carbonate can cause dizziness, headaches, or worse. We set up all decanting and batch mixing in rooms with forced ventilation. Hood fans and automated sensors trigger if vapor readings jump even a little. Not everyone sees the risk until they miss a day of work from exposure, but we remind staff regularly by sharing real field stories where inadequate ventilation led to medical events.
Nothing tests a team like a sudden spill. Our crew relies on well-placed spill trays and correctly labeled absorbents. We avoid drains or open channels where liquid might escape into the wrong place. After a cleanup, used absorbents and PPE go in dedicated chemical waste bins, never general trash. One careless toss can cascade into a bigger hazard, especially in bulk storage zones. We inspect secondary containment every shift—dents, hairline cracks, and corrosion tell us which units need swapping before they fail. If spillage happens, the whole crew drills until everyone knows the response protocol without hesitation.
Methyl Ethyl Carbonate stands out as flammable, with vapors forming explosive mixtures with air. Our warehouse keeps drums away from ignition sources and heat—no smoking signs are not just for show. Every new worker gets a hands-on tour of the bonded storage area, learns how to read labeling, and practices using powder extinguishers. It takes more than a signed signoff sheet to build this vigilance. We scatter temperature sensors through the stacks and double-check ground wires and static bonds before every bulk transfer. Fire drills stay regular, and we treat any lapse as a red flag across departments.
Anyone spending years manufacturing and handling Methyl Ethyl Carbonate sees how quickly an overlooked safeguard flips into a headline incident. Institutional memory—the stories, close calls, and shifted habits—protects more than written guidelines. We build our safety culture from lessons learned hard and shared honestly. Investing in training, upgrading containment, and reinforcing ventilation never shows up as a big news story, but this work keeps our crew healthy and productive every season.