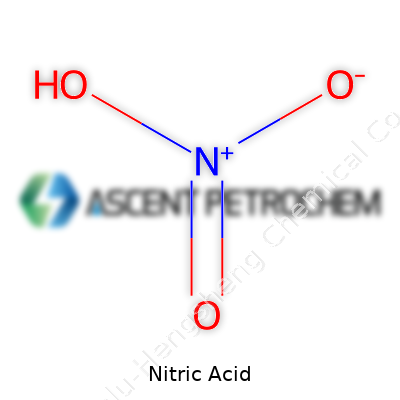
Nitric acid’s legacy stretches deep into the narrative of industrial chemistry. Over the centuries, this potent chemical has carved an indispensable role, originally recognized as aqua fortis in alchemical texts and later becoming a linchpin in mineral acid research. Early processes, such as those using saltpetre and vitriol, placed tight restrictions on purity and scale. The introduction of the Ostwald process, which relies on catalytic oxidation of ammonia, drove modern bulk production, pushed industry forward, and set the benchmark for today’s high-volume, high-purity standards. As manufacturers, we witness how evolutionary leaps in production have translated into reliability in both quantity and quality, especially as demands for electronics, fertilizer, and energetic materials climb.
Nitric acid stands as a clear, colorless liquid under pure conditions. In daily industrial practice, faint yellow or brown hues, tracing to nitrogen dioxide impurities, often appear due to storage or transportation effects. The material brings immediate reactivity and a tendency toward decomposition in light or heat, so drum and bulk tank operations require careful handling. Produced through well-controlled oxidation and absorption, varying concentrations serve different markets, from agricultural ammonium nitrate to high-grade formulations for microelectronics. Every batch tells a story of upstream ammonia integrity, careful catalyst temperature control, and downstream scrubbing to ensure both minimal emissions and maximum product yield.
In our plants, nitric acid emerges with a characteristic sharp odor and remarkable oxidative power. Standard concentrated nitric acid—usually 68-70 percent—boils just above 120°C, yet starts releasing fumes, which deliver both risk and visual cue that containment remains crucial. Miscibility with water ensures easy blending, which puts extra emphasis on vessel material selection; corrosion rapidly attacks ordinary steel, pushing us towards stainless and specific non-metallic alternatives. The acid’s strong oxidizing nature not only sustains its effectiveness in nitration reactions, but also demands strict separation from organics, reducing agents, and combustibles. Over many years, seasoned plant operators learn to navigate nitric acid’s ever-present readiness to react, which, left unchecked, brings equipment damage and injury risk in a blink.
Technical standards drive our process controls, not just regulatory paperwork. Maintaining consistent titratable acid content, trace metal levels, and freedom from contamination takes ongoing analytical commitment. Lot-by-lot analysis for arsenic, heavy metals, and non-volatile matter ensures the outgoing product meets both internal and customer-driven specs. Labeling reflects concentrated, dilute, or fuming nitric acid classification, signaling different hazards and storage needs. We also mark UN number and hazard pictograms according to international transport codes, since small deviations in strength, color, or contamination profile mean cascading complications downstream.
In our facilities, the Ostwald process dominates. Ammonia gas, sourced from either on-site or third-party suppliers, is vaporized, then injected into a platinum-rhodium catalyst bed under tightly regulated temperature and pressure. Oxygen converts ammonia to nitrogen monoxide, which further oxidizes to nitrogen dioxide. Water scrubbing dissolves the gas, outputting aqueous nitric acid of variable strength. Each phase brings critical control points—catalyst efficiency, tail-gas treatment, and absorber function—requiring skilled technicians and constant feedback from in-line and laboratory analyzers. These choices shape both our efficiency and carbon footprint, reminding us every day that clean, reliable production remains an ongoing project.
Few substances command as much technical respect as nitric acid on the reaction floor. In our plants, it plays dual roles: a reagent for oxidation and a nitrating agent for downstream synthesis. High-purity acid feeds explosives manufacture, dye intermediates, and pharmaceutical precursors by introducing nitro groups with selectivity and vigor. Nitrate salts, derived from neutralization with bases, underpin fertilizers worldwide. Reactivity extends to passivating metals, etching semiconductors, and decomposing organic waste in environmental applications. At the same time, nitric acid’s readiness to release nitrogen oxides on contact with organic matter, and its ferocity during exothermic reactions, set high benchmarks for training and process safety.
Nitric acid runs under many aliases in various sectors: aqua fortis, spirit of niter, hydrogen nitrate. In regional trading and bulk sales, you’ll see designations such as fuming nitric acid for higher concentrations or red fuming for product dissolved with nitrogen dioxide. Production runs for electronics, certified low-contaminant blends, and research grades follow distinct nomenclature on paperwork and inventory systems, keeping communication clear from loading dock to laboratory.
Health and environmental rules challenge our manufacturing teams constantly. Nitric acid burns living tissue on contact—a hazard that all site personnel internalize after a single brush against unprotected skin. Fumes corrode respiratory tissues, requiring effective local exhaust, monitored atmospheres, and respiratory protection in hot work and confined spaces. Spills demand more than neutralization routines; runoff threatens waterways, emphasizing our dual job as manufacturer and environmental steward. Regular training covers both everyday PPE routines and acute emergency response, including containment, decontamination, and detailed incident reporting. We log every near-miss, turning mistakes into operational wisdom that sharpens our next shift.
Fertilizer production remains the backbone application, linking factory outputs to global food chains. Nitrates derived from nitric acid give fields the nitrogen charge that crops demand. Beyond agriculture, our product sustains powerful oxidants for the aerospace sector, laser and semiconductor etching for electronics, as well as pigments, surfactants, and even select organic solvents. Each destination brings a different purity requirement, packaging need, and transport regulation, locking nitrogen chemistry to the rhythms of steel foundries, research labs, and recycling yards alike.
Research teams in our company chase efficiency and sustainability under relentless pressure. We push for lower-energy catalysts, better separation and recovery of nitrogen oxides, and ongoing efforts to widen the spectrum of co-products. Investment in process modeling, pilot testing, and analytics continues, directed not just by profit, but also by client requests for trace constituent reduction and low-carbon production. Collaborative efforts with academics and equipment suppliers speed up breakthroughs, whether in abatement of N2O byproducts or development of tailored grades for advanced applications. Smarter control and diagnostics tie into digital platforms, showing that old-line chemistry evolves fastest where tradition and technology meet.
Toxicological research provides the backbone for workable safety limits. Biological monitoring shows that nitric acid uptake impacts upper respiratory health at even low airborne concentrations. Ongoing longitudinal studies probe links to chronic conditions, guiding new exposures and tighter ceiling values for airborne nitric acid. Wastewater studies clarify how acid spills affect aquatic life and help design the next round of process recovery tanks, not just temporary pH neutralizers. Regular health surveillance, not just for production staff but also third-party contractors, forms part of our standing operational review, translating published studies into daily routines.
Market and innovation forces shape our path as demand collides with environmental pressure. As agriculture struggles to feed rising populations, the need for nitrate-based fertilizers persists—yet we’re equally pressed to cut nitrous oxide emissions and rethink conventional ammonia sources. Green ammonia, powered from renewable electricity, stands to transform upstream inputs, offsetting some of the sector’s historic carbon imprint. Research into catalyst longevity, process intensification, and real-time emission capture moves from pilot scale to mainstream every year. We adapt not just to satisfy compliance, but also to create a robust, future-ready platform where nitric acid proves as sustainable as it is indispensable. Every step forward in plant design and training raises new questions, keeping the field dynamic and our engagement rooted in practical experience.
Nitric acid has shaped the modern chemical landscape more than most people realize. Inside our plant, we witness this substance transform raw materials into products that underpin agriculture, tech, health, and beyond. Experience in making nitric acid provides a view rarely shared outside the industry—one built not on guesswork, but on day-to-day realities.
Nearly three-quarters of all nitric acid goes into fertilizer, especially ammonium nitrate. In our facility, we blend this into granules farmers rely on for maximizing their yields. That matters for global food stability; the world depends on high-output farming because arable land per person keeps shrinking. Efficient use of nitric acid in the nitrophosphate route gives us more precision with nutrient blends, reducing waste and improving both crop and environmental outcomes. Our manufacturing choices—like controlling reaction temperatures and purity—directly affect solubility and safety in the final fertilizer.
Nitric acid remains central in making explosives. Ammonium nitrate from our reactors finds its way into mining operations and infrastructure projects. Safety is always on our mind—we invest in online monitoring systems that help prevent runaway reactions and contamination. This keeps both our workers and clients safe. The reliability of our product sets the standard for blasting performance and reduces unwanted environmental side effects.
Working with nitric acid goes far beyond bulk production. In metal refining, controlled amounts of nitric acid strip impurities from copper, silver, and gold. We pay close attention here, as stronger solutions can attack both base and precious metals, leaving nothing behind if poorly managed. For electronics, high-purity nitric acid supports etching circuits and cleaning silicon wafers. This is not a place to cut corners—minor contaminants in the acid may ruin entire production batches of microchips. Because of this, we keep our facilities up to the demanding standards the sector requires, investing in extra purification steps.
Environmental compliance demands as much ingenuity as raw chemistry. On-site, nitric acid plays a key role in wastewater treatment, oxidizing harmful materials before we release anything. This step prevents heavy metals and nitrogen compounds from making their way into rivers and lakes. Our operators remain vigilant, monitoring effluent streams and adapting practices continuously as regulations evolve.
As one of the building blocks of modern chemistry, nitric acid supports the synthesis of medicines, dyes, and specialty chemicals. Our reactors see batches for active pharmaceutical ingredients rolling through side by side with pigment intermediates. Flexibility matters just as much as throughput; switching between applications without cross-contamination only works with careful planning and proven cleaning protocols.
In manufacturing, price and purity mark the difference between success and disaster. Over the years, we’ve dealt with volatility in energy costs, changes in raw material supply, and mounting environmental regulations. Continuous investment in catalyst optimization and heat recovery lessens both the cost and the carbon footprint of our operation. Nitric acid production demands careful stewardship, responsibly handling risks alongside the industry’s essential role in growth, food security, and innovation.
Everybody who steps foot on our shop floor recognizes the bite of nitric acid. You spot that yellowish fume from fifty feet away, and your skin or lungs never forgive you for skipping personal protective equipment. Eyes demand tight chemical goggles; faces call for full shields; hands require acid-resistant gloves. We go with thick rubber or neoprene, something that stands up when the acid tries to seep through. Friend of mine once got a tiny splash on a bare patch of wrist, and the burn humbled him for weeks.
Nitric acid doesn't hide its intentions. It fumes aggressively, especially under warm conditions, quick to fill a space with nitrogen dioxide. Our plant runs with serious scrubbers and exhaust systems that move air fast. Workers know to never open containers except under ventilation hoods. The difference between a tight chest and a clear set of lungs often comes down to fan speed and extractor placement.
Every bottle and drum on our line uses materials that stand up to acids, never old steel, never containers with suspect linings. We go with glass, certain plastics, or acid-resistant alloys, no exceptions. If a container cracks or leaks, nitric acid finds its way onto metal and reacts, releasing toxic gas. Over a decade of moving this product, nothing causes a panic like the hiss of nitric acid on the wrong material.
No one rushes with nitric acid. Any experienced chemist or operator will double-check every hose, every gasket, every valve before charging a tank. Spills come from quick hands, not careful ones. Our loading stands and transfer points keep containers stable, and two sets of eyes watch every critical pour. We train new employees by partnering them with veterans who remember every close call.
If something spills, crews pour on sodium bicarbonate or lime right away. Water alone doesn't help; it just spreads the mess. Several times we've seen folks from other industries try water first, only to watch the problem multiply. The right neutralizer takes the sting out of nitric acid, so we stock bins of bicarbonate at every handling zone.
Nitric acid brings out the worst in many substances. Organic materials, oils, even some metals react fiercely—sometimes with heat, flame, or poison gas. Our team reviews every process change and bans casual mixing. Anybody who’s handled it for a year has seen what a drop in the wrong place can do to a workbench or a chemical tote.
Years on the shop floor teach one lesson about nitric acid: respect grows with experience. We rely on rigorous training and clear procedures, but it’s the collective memory of past incidents that makes everyone vigilant. Manufacturing doesn’t forgive shortcuts with this chemical. Detail and discipline remain the best insurance we have.
Nitric acid runs through countless chemical plants, fertilizer factories, and metal finishing shops. In our plant, we face its challenges head-on. At high concentrations, nitrogen oxides and that sharp, acrid fume turn possibility into danger. It eats through common steel and attacks organic materials. Years in production taught our team that casually stashing nitric acid on a regular shelf or in a standard drum leads straight to equipment damage, worker risk, and costly downtime. Acid that seeps, weeps, or vaporizes spells trouble fast.
Stainless steel, grades with enough chromium and molybdenum to hold firm, earns trust for storage tanks. Polyethylene and certain fluoropolymer linings also handle the job for low concentrations. Experience rules out most other plastics and metals. We once learned the hard way that aluminum, copper, or brass turn into a corroded mess in no time. Piping, valves, and gaskets—one weak component leads to breakdown, so the whole chain takes the same careful approach as the main tank.
Ventilation splits the line between routine work and emergency. Nitric acid gives off choking fumes, especially if it heats up or meets incompatible materials. Strong airflow, kept away from busy walkways, helps protect everyone in the area. Direct sunshine turns tanks and containers into hot spots, building pressure and increasing fume release. We place every storage vessel in a shaded, temperature-stable spot. Acid never lives near bases, reducing agents, or organic stock. One bad mix can generate brown fumes or violent reactions—no shortcuts taken here.
Tanks and transfer lines earn routine checks. Over years, even stainless steel gets pinholes or weakening welds. Acid-resistant coatings peel or bubble if ignored. Each refill brings a chance to spot early warning signs. Leak sensors deserve a place beneath every main vessel. Drip trays and bunded floors catch anything escaping the main tank. From personal experience, small leaks go unnoticed until a cleanup turns major—good barriers keep a minor issue from turning into an emergency.
No matter how good the setup, untrained hands can defeat it. Every shift in our plant brings another reminder—use the right gloves, splash goggles, chemical-resistant aprons. We insist on strict routines for decanting and sampling. Fast transfers tempt skips in procedure, yet the price for a shortcut is too high. Evacuation routes, emergency washes, and backup neutralization agents belong in every storage plan. Respect for nitric acid demands steady focus, not just convenient storage space.
Best practices found in protocols only make sense once tested on the real factory floor. From plant managers to line operators, first-hand experience shapes the design, layout, and ongoing care of our nitric acid stores. Industry guidelines help, but daily use, maintenance, and occasional surprises teach lessons that stick: treat nitric acid storage as a living process, not a checked box. That philosophy safeguards our workers, product quality, and the site itself.
Customers come to us with straight questions about concentration. They want numbers with an explanation, not just comfort words pulled from brochures. Nitric acid often arrives at 68% concentration. Some ask what this number means for them. It means real performance, just not the same punch as so-called “red fuming” or “white fuming” nitric acid above 95%. Bulk of industrial work leans on the 67% to 68% grade. This covers metal finishing, fertilizer, and other chemical synthesis.
Nitric acid draws water from air. What leaves our plant at 68% stays stable in sealed drums. Pour it into open tanks or pump through leaky lines and the balance can shift. End use matters. Some plants can work with small swings in strength, others can’t. A fertilizer plant wants reliable action during neutralization or reaction. A metal etching operation needs repeatable results on every run; changes in dilution can lead to ruined parts.
Getting the percentage right affects the entire operation. If the nitric acid slips under 67%, many reactions slow down or fall outside spec. If it climbs toward the 95% mark, equipment corroded by excessive acid vapor doesn’t last long. Engineers in the field learn to ask for numbers, test them, and keep their own records because surprises waste money and, sometimes, damage systems.
Pure numbers keep customers honest, too. Mixing processes rely on facts, not trust. In high-volume applications, switching from a 68% supply to 65% or 71% might throw off a whole day’s production. This is not about theory. In our years of monitoring shipments, correcting drift and responding to out-of-spec reports, we see what happens when end-users expect one strength and get another.
Industrial nitric acid comes freshly distilled from ammonia or nitrogen oxides. As a manufacturer, we sample each lot directly from the process stream. Acid titration in our lab gives an accurate reading, confirmed with in-process sensors before filling bulk tankers. Routine checks catch changes fast: if a batch slides away from our customer’s spec, we hear about it. Every error costs us time and reputation.
Over decades, we have invested in inline monitoring, not just batch titrations. Sensors alert the shift crew far sooner than old manual methods. In uncontrolled settings—even a warm storage yard—water vapor finds a way in, so strength drops. That’s why acid gets shipped and stored in special containers. Every drum or tanker loaded at our warehouse holds a printout of the test result. Buyers start their assessment with a number they can verify.
Concentration drift is a reality across the industry. Whenever possible, we suggest minimizing exposure to air, checking seals on storage tanks, and sampling the acid before adding to a critical process. If a batch turns out weaker than the recipe calls for, plant managers can adjust the dosages, or in extreme cases, blend with higher concentration acid. Retesting before using acid is always cheaper than cleaning up after an unexpected outcome.
Manufacturing nitric acid to a specific concentration is the easy part. Maintaining its strength until it goes into a reactor or onto a production line means working with customers, not just shipping barrels. Open communication, scheduled maintenance, and periodic retesting keep production moving and help everyone sleep better at night. It’s not just chemistry—it’s trust built on real numbers.
People working at the reactor face a simple reality: not all nitric acid is equal. From my experience on the production floor, different jobs call for distinct grades, and cutting corners on purity creates big problems downstream. Nitric acid moves from high-purity electronics to heavy-industry nitration lines. This isn’t just about labels. Every grade comes with its own set of challenges, costs, and safeguards.
The nitric acid most factories want for general chemical synthesis sits around 68% concentration. This technical grade easily handles routine processes—fertilizer manufacturing, basic metal treatment, and standard chemistry. Factories with less margin for error in their results—think pharmaceutical or semiconductor outfits—seek so-called “high-purity” or “ultra-pure” grades, where trace metals and organic contaminants might disrupt a sensitive process or cause equipment corrosion. Achieving this means production teams deploy extra steps: fine filtration, custom distillation, and rigorous lot tracking become the rule rather than the exception.
On the other end of the spectrum, dilute grades exist for specific uses like etching, cleaning, or lab preparation. Each concentration demands different stabilization chemistry and tankage, since pure nitric acid decomposes on exposure to light and heat, forming toxic brown fumes. Workers on site know this isn’t just theory—the stench and color let you know immediately if the concentration drifts.
Customer requests drive these purity differences. An engineer troubleshooting a stoppage on an electronics line will lose patience fast with acid ‘good enough’ for fertilizing fields. Even small lapses in chloride or iron levels can bring million-dollar losses in high-precision manufacturing. In fertilizer and explosives, on the other hand, minute impurities matter less, but cost and volume rule every decision. Often, these customers set their standards not to chase zero contamination, but to high enough quality for their needs, at a price they can defend.
Keeping track of grades at the plant isn’t a paperwork game. Tanks, pipes, and pumps must get flushed and validated between batches; failure can mean years of trust lost with a single batch shipped to the wrong customer. Sampling and analysis aren’t glamorous tasks, but catching a contamination early saves a headache later. Generating the highest purity means running smaller campaigns, losing time to changeover, and investing in people trained to spot problems before they hit specification sheets.
Manufacturers learned the hard way that standardized grades matter. Over the years, working with auditors and quality managers, we built batch tracking, contamination control, and closed-loop feedback from customers into our routines. Instead of chasing a one-size-fits-all product, it pays to build flexibility into the plant—dedicated lines for ultra-pure, bulk infrastructure for lower grades, and clear communication with every customer about the grade supplied.
The push for cleaner, more reliable nitric acid comes not from marketing, but from hard lessons in production. Every order shapes how we set up, sample, and ship the next one. That’s what keeps customers coming back. It isn’t just nitric acid; it’s trust in every drop.