Toluenediamine has built its legacy step by step, rooted in the classic age of organic chemistry, with research tracing back to late 19th-century aromatic amine developments. As a chemical manufacturer operating for decades, we have seen firsthand how toluenediamine earned its position as a foundational intermediate in material science and polymer development. As demand for high-performance polyurethane foams and engineering resins increased across the globe, so did the investments and focus on refining the routes for toluenediamine production, both to improve purity and ensure higher consistency in each batch. The shift from small-scale synthesis to large-volume industrial production challenged early processes, pushing developers to adapt aromatic amination and catalytic hydrogenation technologies, making the process safer and output more dependable. At our own facilities, switching from batch reactors to safer, continuous systems marked a turning point. Emphasis on robust process controls, integrated with emissions-cutting strategies, has drastically improved yield and reduced by-product streams compared to early practices.
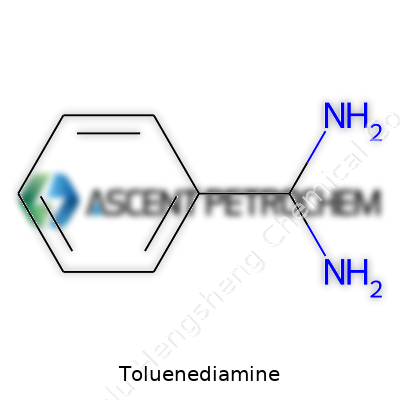
Toluenediamine, often referenced as TDA, typically refers to toluene-2,4-diamine and toluene-2,6-diamine, both commercially important due to their role in polyurethanes. The industrial form is often a mixture, produced from dinitrotoluene by catalytic hydrogenation. TDA distinguishes itself through two active amino groups on a methylated benzene ring, a structure offering reactivity while maintaining workable stability. Over the years, our teams learned to appreciate subtle differences between the isomers in behavior and downstream reaction rates. Because TDA displays notable reactivity toward isocyanates, it has shaped the evolution of polyurethane chemistry, giving formulation chemists tremendous control over the physical properties of the polymers they design.
Our process engineers and laboratory staff are intimately familiar with TDA's sharp, amine-like odor, and its pale to amber appearance in solid or liquid form, depending on storage conditions. Melting points for the pure isomers hover around 99°C and 63°C. TDA dissolves in common polar solvents, such as alcohols and ethers, and its high boiling point suits high-temperature polymer synthesis. Reactivity with oxidants, acids, and halogenated compounds means the storage environment must be strictly controlled. Avoiding copper or brass fittings, for instance, became standard practice early on. Repeated exposure to humidity or high heat can promote secondary reactions or degrade the product, so inventory is rotated and checked regularly.
With so many technical fields relying on TDA as a primary component, close attention to specifications has always mattered on our production lines. Purity levels above 98%, low moisture content, and a well-controlled isomer ratio form the backbone of our outgoing batch certifications. Trace impurity testing, especially for aniline, nitrotoluenes, and heavy metals, gets prioritized not only to meet regulations but to guarantee consistent downstream performance for our industrial partners. Labeling now highlights hazard classes—acute toxicity, skin sensitization, aquatic hazards—based on evolving scientific consensus and regulatory obligations. Because TDA’s toxicity is well-documented, transparent communication with customers plays a real role in safe usage.
Our industry long ago settled on reductive hydrogenation as the most reliable method for manufacturing TDA at scale. Dinitrotoluene (DNT), itself a product of nitrating toluene, undergoes catalytic hydrogenation in dedicated reactors using sponge nickel or modified platinum group catalysts. Controlling temperature and pressure makes the difference between high-purity yield and complications like over-reduction or ring hydrogenation. We have invested over the years in in-line analytics—GC and HPLC monitoring—to catch issues in real time. Contamination from catalyst poisons or process upsets used to pose obstacles. We now mitigate these risks through closed-loop controls and routine maintenance. Waste by-products demand careful management; separation and destruction of side streams have improved, but maintaining operator vigilance is essential.
Much of the excitement in TDA chemistry revolves around its reactivity with phosgene and isocyanates. Polyurethanes, especially flexible and rigid foams, emerge when TDA reacts with diisocyanates like TDI. Reaction speed, foam porosity, and resilience depend on the specific ratios of TDA isomers, which prompts us to tailor blends according to customer needs. Beyond that, TDA’s amine groups allow for further derivatization—acylation, alkylation, or sulfonation—all strategies for introducing specific functionalities in specialty polymers or intermediates. In our R&D labs, ongoing work investigates how these modifications might tune properties like dye uptake or chemical resistance in end-use applications. Side reactions—oxidation, nitrosation—are always present in background, but decades of analytical tracking have taught our staff methods for anticipating and neutralizing unwanted pathways.
Names for TDA and its isomers—2,4-diaminotoluene, 2,6-diaminotoluene, TDA, toluylenediamine—create confusion even among scientists and procurement professionals. Our documentation always cross-references CAS numbers and common trade names to minimize mistakes, because mislabeling can bring serious safety and application risks. In international markets, translation differences add extra complexity, so our export teams verify product coding and approve labeling before shipments move. Communication with customers and partners about synonyms, purity grades, and form (liquid, solid, blend) remains a daily aspect of our quality operations.
On our plant floor, safety dictates every decision involving TDA. Employees quickly learn to treat the material with respect. Exposure causes skin sensitization, potential mutagenicity, and acute toxic effects if inhaled or ingested. Closed handling systems, local exhaust, and full PPE for operators reflect our hard-won experience. The temptation to cut corners never outweighs the consequences, so our safety audits set strict protocols for all process stages. Spill containment, effective training, and correct first aid administration all matter. Over years, even short-term exposure at low levels leads to cumulative health risks. This reality shapes every handling, packaging, and transport practice at our facilities. We cooperate with insurers, health and safety authorities, and our own teams to update risk assessments year over year.
Polyurethane production stands out as the top driver for global TDA consumption, impacting industries from home furnishing to automotive seating and insulation. Flexible polyurethane foams, rigid boards, elastomers, and coatings draw directly on TDA’s chemical backbone for their structural integrity and resilience. In addition, some dyes, pigments, and rubber processing chemicals trace origins back to TDA intermediates. Custom surfactants, specialty polymers, and even certain explosive compounds count aromatic diamines as essential synthesis steps. Throughout, our technical advisors consult closely with customers on formulation optimization, troubleshooting, and regulatory compliance issues that emerge during final product development.
Scientific curiosity keeps pushing boundaries with TDA, and as manufacturers, we maintain active research programs tracking several fronts. Process engineers look for routes that use green chemistry, examining catalytic systems that reduce energy demand or lower greenhouse gas emissions. Ongoing collaborations with universities and research institutes aim to develop safer analogues, address impurities, and increase functional diversity for advanced applications. In our labs, computational chemists model reaction mechanisms to guide scale-up of new processes, while formulation scientists study the impact of minor isomeric changes on polyurethane foam quality. Innovations in trace analysis—mass spectrometry, NMR techniques—give us better tools for understanding side reactions and impurity sources, sharpening our competitiveness.
Decades of published studies underscore the significant toxicity of both TDA isomers. As operators and stakeholders, we have witnessed the direct effect of workplace exposures and the burden on downstream users managing residual TDA in waste streams. Universal adoption of strict occupational exposure limits—far tighter than in the 1980s—reflects the scientific consensus on hazards ranging from skin sensitization to carcinogenicity. Our own site history includes investments in engineering controls to keep air and surface contamination below threshold levels, as well as medical surveillance and reporting protocols for all involved personnel. Newer research investigates metabolic pathways, seeking to identify less-hazardous structural analogues. Wastewater and emission controls have tightened considerably as the industry matures. This focus on worker and environmental health no longer competes with productivity; both go hand in hand.
The outlook for TDA remains intertwined with global polymer demand trends, particularly for high-performance foams and specialty elastomers. Driving forces include regulatory pressure for lower-toxicity alternatives and demand for greener processes. Within our industry, efforts now focus on improving hydrogenation catalyst lifetimes, reducing process emissions, and pushing energy efficiency gains. Chemists keep testing novel routes that use bio-based feedstocks, aiming to offer equivalent functionality with reduced environmental cost. The adoption of circular economy practices—recovery, reuse, and recycling of aromatic diamine-containing materials—marks an area attracting fresh investment and public attention. As new toxicity data emerges, we anticipate further tightening of exposure limits, stricter waste controls, and a possible shift toward safer derivatives in some regions. Through ongoing collaboration with academic and industrial partners, we plan to remain at the forefront as regulations, markets, and technologies evolve.
Fresh batches of toluenediamine leave our reactors every week. If you trace those molecules, they don’t rest on a storeroom shelf or disappear into the ether. They ride out into the world’s supply chains, at the center of complex chemistry that shapes everyday products. Our chemists have worked with this raw material for years, and the story keeps unfolding as old challenges meet new needs.
Look at the seat you’re sitting on, the insulation in your walls, the vibration-damping pads in your appliances. All of these might owe their performance to polyurethanes, and polyurethanes rarely exist without a key catalyst: toluenediamine. Factories large and small depend on its consistency—batch after batch—because making rigid, lightweight foam with precise performance takes stable, refined input. Small fluctuations in quality multiply through the production line, opening the door to faulty products and costly downtime. Our job starts with managing purity, keeping color and moisture in check, and running trusted analytical checks before every shipment leaves our plant. Defects in the backbone molecules mean defects in the final piece.
Vehicle manufacturers and equipment builders keep painting and powder-coating lines busy year-round. They want coatings that can withstand high temperatures, resist solvents, and brave the weather. Isocyanate-cured epoxies and polyurethane coatings give them that durability. Our toluenediamine provides a crucial hardening component, cross-linking resins for that tough finish. Minor shifts in composition or impurity levels can change curing times, surface appearance, or corrosion resistance. Years of direct feedback from end users press us to put special care in every drum.
The waxy blocks that leave our storage tanks sometimes go into more specialized chemistry. In pharmaceuticals, scientists use toluenediamine as a synthetic intermediate—the raw foundation for several molecules in development. The scale is much smaller than in foams or coatings, but the expectations are even sharper. We’ve invested in additional purification steps and dedicated storage to prevent contamination, since even trace impurities can derail a research project or fail a clinical trial. Recent work on advanced dyes also draws on toluenediamine’s unique reactivity to create vibrant, stable colors for lab and industrial use.
Handling large volumes of aromatic amines comes with hazards you don’t see outside the plant fence. Toluene-derived diamines demand careful containment, with strict ventilation, personal protective equipment, and thorough training for every employee. Regulatory scrutiny has increased every year, pushing us to improve our emissions and waste processing. We see open communication with workers and regular reviews as key tools—not just rules for compliance, but for fostering a safer workplace and surrounding community.
We keep collaborating with polyurethane and coating producers to reduce waste, reclaim material, and adapt our processes as industry and environmental standards climb. Circular economy initiatives nudge us toward more efficient resource management, closing loops where possible and searching for less hazardous substitutes when effective.
The demand for toluenediamine isn’t fading. High-performance foams, coatings, and specialty chemicals all lean on the reliability and reactivity of this compound. As more regulations and expectations arise, manufacturers like us face twin pressures for efficiency and safety. Each day in the plant, the direct relationship between careful production and trusted application keeps us focused—turning chemical knowledge and operational experience into practical solutions for manufacturers worldwide.
Every morning brings us into direct contact with raw chemicals—powders and liquids few people outside industry ever see. Toluenediamine (TDA), which we handle for polyurethane production and dye manufacture, has a profile that always pulls discussion around safety. Putting aside theoretical discussions, let’s talk from the shop floor and lab experience as a chemical manufacturer.
We see real risks with TDA exposure. Inhalation of vapors or dust, or prolonged skin contact, highlights risks most industry workers already know or find out quickly. Short-term exposure can irritate eyes, nose, and throat. Our safety team documents skin irritation ranging from mild redness to more severe dermatitis. Incidents where workers needed medical attention have always pushed us to improve local ventilation and personal protective equipment protocols.
Long-term concerns go deeper. Decades-old studies, echoed by regulatory authorities, show that TDA can cause genetic mutations in animals and signals potential carcinogenic effects in humans. Although direct cancer risks for humans remain under study, laboratories treating TDA with caution are on the right track. It does not justify panic, but it demands respect.
In our plants, practical control matters more than policies on paper. Ventilated enclosures around points where powders are transferred or solutions are made keep airborne levels low. Respirators and gloves are worn not as an optional inconvenience, but as standard. When a drum spill occurs—and it will, despite the best plans—the team responds with a focus on minimizing exposure. Decontamination areas, specialized absorbents, and detailed scripts for evacuation and cleanup protect everyone down the line.
Newcomers quickly learn the value of these measures after their first close encounter with TDA dust. The chemical reacts slowly with air and moisture, but its vapors can hang around, especially in enclosed spaces. Our monitoring equipment picks up spikes, signaling us to adjust processes or check for leaks before people feel symptoms.
TDA falls under close scrutiny by occupational regulators across markets. Permissible exposure limits exist to cap the airborne concentrations workers can encounter over a shift. Meeting these benchmarks is not just compliance—it keeps our people healthy and production lines running without unexpected losses.
Training works as a frontline defense. Before stepping onto the plant floor, everyone studies potential health impacts and learns precise handling routines. We rotate job assignments, so no single worker faces elevated risk for extended periods. Routine medical checks let us spot early signs of overexposure, with health surveillance woven into our operating rhythm instead of being a distant afterthought.
Process engineering continues to chip away at risk. Automated weighing and closed-loop transfer systems keep human contact with raw TDA to a minimum. For specialty applications that cannot yet substitute TDA, high-integrity containment technology aims to all but eliminate leaks and spills. Industry talk sometimes points to new amine blends with better toxicological profiles, but replacement rarely proves simple.
Those of us running reactors or cleaning up spills know TDA has sharp edges. By leaning into modern safety systems and workplace transparency, manufacturers can keep risk controlled, health protected, and lessons from the past always close at hand.
After decades spent working on the production line and managing the day-to-day issues that surface in chemical manufacturing, I’ve learned that the physical properties of a compound like toluenediamine directly shape its performance in real-world applications. Nothing drives this point home better than watching a batch go off spec because of a misunderstood phase change or a careless mishandling of the material at the wrong temperature. Those details that look dry on paper end up dictating costs, safety measures, and what downstream users can achieve.
Toluenediamine typically arrives in granules or crystalline powder. I’ve seen older batches clump together if stored in humid environments, making handling much trickier on the shop floor. The color usually ranges from light tan to gray-brown. Occasionally, poorly controlled syntheses put out off-color batches, and the difference is hard to explain away to auditors or customers who expect consistency. Small details like that reflect on the pride or care put into every part of the operation.
This compound has a melting point that sits just above room temperature, around 99 to 103°C, depending on the isomer. This low melting behavior makes it straightforward to liquefy for feed into reactors—no specialized heating equipment is needed, but if the storage warehouse isn’t insulated, you’ll wind up with a solid block inside your drums during colder months. One particularly harsh winter, I had to supervise teams with industrial heaters for three days just to get the product pumpable—those sorts of practical challenges stick with you and influence decisions on how to break up lots for delivery or which containers to use.
Odor is another point worth mentioning. Even trace vapor escapes during handling release a sharp, amine-like smell—no mistaking it. Technicians working long hours beside open containers know ventilation isn’t just a box to tick for compliance; it’s what makes the difference between a manageable work environment and headaches for half the team. Many newer hands who come from less hands-on roles never forget their first shift with toluenediamine.
These amines absorb moisture fairly easily from ambient air. This aspect poses real storage and quality problems. Moisture uptake could lead to product lumping or, in a worst-case scenario, gradual degradation. A consistently dry warehouse, well-sealed packaging, and frequent QC checks have saved my team more than once from customer complaints—these habits are easy to overlook until the day an unexpected lot recall lands on your desk.
The density, just over 1.1 g/cm³, brings its own impact for both shipping and batching. We constantly need to calibrate pumps and mix tanks, since even a small overestimation multiplies fast when the daily volume climbs. If the product was twice as dense, forklift operators would double their caution—and if it was less, vibrations in transit could mean more container breakages.
Most people outside of chemical production don’t think about how the physical form, phase behavior, and even scent of a material play into plant scheduling and worker safety. Those of us who work with these compounds daily do not have the luxury of abstract curiosity. Every property, down to the feel of powder slipping through gloved fingers, connects to the realities of safe, efficient manufacturing.
Across our production lines, toluenediamine has stood out for both its value and its hazards. Chemists and technicians develop a healthy respect for its potential risks—especially skin and respiratory exposure—after working with it day in and day out. We’ve seen, through real incidents and years of careful practice, what works and what goes wrong. Cans leak; drums warp in summer heat. Gloves wear thin. Steel toes and fitted goggles save more than just time.
Toluenediamine does best in a temperature-controlled, well-ventilated room with zero exposure to direct sunlight. Structural changes and impurities become real problems if product sits somewhere hot or overly humid, especially during long-term storage. A shaded warehouse with sealed concrete floors stops most risks in their tracks. Every new batch, we tighten drum lids and double-check for moisture. Even a trace of water kicks off slow, unwanted reactions. On busy receiving days, we stack containers on chemically compatible pallets—nothing corrosive, no pressure from above.
There’s no substitute for good personal protective gear. Protective gloves and goggles sound like basics, but complacency has caused incidents at many facilities. We enforce a firm line: no access to handling zones without proper gear, full stop. Splash-resistant aprons and face shields earn their spot on the rack. Any time we switch batches or clean up a leak, extra vigilance goes a long way. After one near miss, we changed up our spill containment routines—moving from open floor drains to chemical-resistant spill kits at every access point. Smaller leaks stay small with the right barriers.
Few substances teach respect for ventilation like toluenediamine does. We angle exhaust hoods and open up vents, keeping fresh air circulating through any handling areas. Over the years, we found that stagnant air leads to irritation, so we mapped out fan placement and air exchange rates based on feedback from staff working the late shifts. Metered transfer pumps cut down worker exposure during unloading and mixing—not worth taking shortcuts. Spills and dust get vacuumed up with HEPA filters. Open flames or static sparks stay far from toluenediamine zones. We learned to shut down power tools nearby during transfer.
Real safety goes beyond laminated posters. Every person in our plant, from the new shift tech to the experienced supervisor, gets hands-on practice with emergency routines. The goal is muscle memory—quick action, no panic. After a few close calls, we updated our response plans and ran drills until everyone handled mock spills without hesitation. The best safety policies draw from lived experience, not just checklists. Feedback flows both ways, with anyone able to call out potential weak spots before they become real problems. That approach keeps both people and product safe.
Years of production taught us that reading labels, wearing gear, and following procedures all matter—but constant attention and a culture of learning keep trouble at bay. Each employee holds a stake in safe outcomes. Storage conditions never stay static, and staff turnover brings new challenges. Ongoing education and open discussions about near misses create an environment where everyone feels responsible for improvement. It’s not enough to store and handle chemicals; companies need to invest in systems and people that keep them safe. From our vantage point, experience on the factory floor shapes what’s practical, and what’s essential, more than any outside instruction ever could.
Anyone who’s spent years around toluenediamine knows the sweet, slightly acrid smell isn’t just a warning — it’s a reminder of what this aromatic amine can do to skin and lungs. Daily work around these tanks and reactors gives you a deep respect for every splash and vapor. Nobody in the plant stays careless for long. Breathing in too much can irritate the nose and throat fast, and even small spills on bare skin turn into red, itchy patches before you finish your shift. You learn quickly: gear up if you value your health.
Toluenediamine doesn’t play nice with bare hands. We’ve tested a range of gloves — nitrile and butyl rubber hold up much better than latex, which softens and lets chemical seep through. Thicker gloves are tougher to work in, but there’s no benefit to speed if you lose a layer of skin to a careless drip. Full sleeves, cuffs tucked inside, keep splashes off the forearms. Exposed wrists are a common problem, and tape around the cuffs stops leaks that creep in over a long shift.
Too many learn the hard way that ordinary glasses only protect from flying chunks, not vapors or fine mist off a drum. We stick to tight-fitting chemical splash goggles. It only takes one mistake with a pump hose to send a fine spray to your face. Face shields go on for the big batch transfers or drum changes, especially if you’re working overhead. Chemical burns to the eye put folks out for weeks — nobody wants to be next in line at the eyewash station.
Fans and fume hoods cut the risk, but loading or unloading concentrated toluenediamine stirs up fumes that linger at breathing height. If airborne levels go up, we use organic vapor respirators fitted with the right cartridges. Without the right mask, you taste and smell the chemical before it shows up on the monitor. Respirators fit poorly on anyone with a beard, and training makes a difference — you know it’s working when you don’t gag as soon as you open a pail. Keeping facial hair trimmed and checking seals is as routine as clocking in.
Not every job in the plant calls for full-body coveralls, but mixing or sampling near open drums means splashes are almost certain. Chemical-resistant aprons and long-sleeved coveralls stop most contact before it soaks through to skin. Changing out of work gear before heading home is a habit built from real stories, not just rules on a poster. Wearing contaminated clothing in the break area can cause skin rashes or even bring the chemical to family members. We set aside designated lockers for clean and dirty gear — cross-contamination is a real problem, and nobody wants to pack extra risk for themselves or their kids.
No single piece of personal protective equipment can do it all, but together, well-chosen gear offers strong defense when backed by habits formed in real plant work. Easy access to washing stations, regular gear checks, and a culture of looking out for each other — these aren’t just regulations. They’re grounded in experience, and they keep workers heading home safe day after day. Listening to seasoned operators, updating training when processes change, and treating every leak or splash seriously make the difference between a scare and a serious injury. That’s not just policy; it’s what keeps people confident enough to show up through years of production face to face with chemicals like toluenediamine.